Seasonal malaria vaccination: protocol of a phase 3 trial of seasonal vaccination with the RTS,S/AS01E vaccine, seasonal malaria chemoprevention and the combination of vaccination and chemoprevention
- PMID: 32933955
- PMCID: PMC7493088
- DOI: 10.1136/bmjopen-2019-035433
Seasonal malaria vaccination: protocol of a phase 3 trial of seasonal vaccination with the RTS,S/AS01E vaccine, seasonal malaria chemoprevention and the combination of vaccination and chemoprevention
Abstract
Introduction: Seasonal malaria chemoprevention (SMC), with sulphadoxine-pyrimethamine plus amodiaquine (SP+AQ) is effective but does not provide complete protection against clinical malaria. The RTS,S/AS01E malaria vaccine provides a high level of protection shortly after vaccination, but this wanes rapidly. Such a vaccine could be an alternative or additive to SMC. This trial aims to determine whether seasonal vaccination with RTS,S/AS01E vaccine could be an alternative to SMC and whether a combination of the two interventions would provide added benefits.
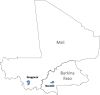
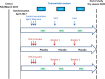
Methods and analysis: This is an individually randomised, double-blind, placebo-controlled trial. 5920 children aged 5-17 months were enrolled in April 2017 in Mali and Burkina Faso. Children in group 1 received three priming doses of RTS,S/AS01E vaccine before the start of the 2017 malaria transmission season and a booster dose at the beginning of two subsequent transmission seasons. In addition, they received SMC SP+AQ placebo on four occasions each year. Children in group 2 received three doses of rabies vaccine in year 1 and hepatitis A vaccine in years 2 and 3 together with four cycles of SMC SP+AQ each year. Children in group 3 received RTS,S/AS01E vaccine and four courses of SMC SP+AQ. Incidence of clinical malaria is determined by case detection at health facilities. Weekly active surveillance for malaria is undertaken in a randomly selected subset of children. The prevalence of malaria is measured in surveys at the end of each transmission season. The primary endpoint is the incidence of clinical malaria confirmed by a positive blood film with a minimum parasite density of 5000 /µL. Primary analysis will be by modified intention to treat defined as children who have received the first dose of the malaria or control vaccine.
Ethics and dissemination: The protocol was approved by the national ethics committees of Mali and Burkina Faso and the London School of Hygiene and Tropical Medicine. The results will be presented to all stakeholders and published in open access journals.
Trial registration number: NCT03143218; Pre-results.
Keywords: epidemiology; immunology; tropical medicine.
© Author(s) (or their employer(s)) 2020. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: None declared.
Figures
References
-
- BACKGROUND PAPER ON THERTS,S/AS01 MALARIA VACCINE, 2015. Available: https://www.who.int/immunization/sage/meetings/2015/october/1_Final_mala...
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous