Adeno-associated virus serotype 1-based gene therapy for FTD caused by GRN mutations
- PMID: 32937039
- PMCID: PMC7545603
- DOI: 10.1002/acn3.51165
Adeno-associated virus serotype 1-based gene therapy for FTD caused by GRN mutations
Abstract
Objective: Dominant loss-of-function mutations in the gene encoding the lysosomal protein, progranulin, cause 5-10% of frontotemporal dementia cases. As progranulin undergoes secretion and endocytosis, a small number of progranulin-expressing cells can potentially supply the protein to the entire central nervous system. Thus, gene therapy is a promising treatment approach.
Methods: We evaluated adeno-associated viral vector administration into the cerebrospinal fluid as a minimally invasive approach to deliver the granulin gene to the central nervous system in a murine disease model and nonhuman primates.
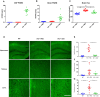
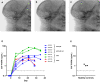
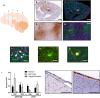
Results: In progranulin-deficient mice, vector delivery into the lateral cerebral ventricles increased progranulin levels in the cerebrospinal fluid and normalized histological and biochemical markers of progranulin deficiency. A single vector injection into the cisterna magna of nonhuman primates achieved CSF progranulin concentrations up to 40-fold higher than those of normal human subjects and exceeded CSF progranulin levels of successfully treated mice. Animals treated with an adeno-associated virus serotype 1 vector exhibited progranulin expression fivefold higher than those treated with an AAV5 vector or the AAV9 variant, AAVhu68, apparently due to remarkably efficient transduction of ependymal cells. Progranulin expression mediated by adeno-associated viral vectors was well tolerated in nonhuman primates with no evidence of dose-limiting toxicity, even at vector doses that induced supraphysiologic progranulin expression.
Interpretation: These findings support the development of AAV1-based gene therapy for frontotemporal dementia caused by progranulin deficiency.
© 2020 The Authors. Annals of Clinical and Translational Neurology published by Wiley Periodicals LLC on behalf of American Neurological Association.
Conflict of interest statement
J.M. Wilson is a paid advisor to and holds equity in Scout Bio and Passage Bio; he holds equity in Surmount Bio; he also has a sponsored research agreement with Ultragenyx, Biogen, Janssen, Precision Biosciences, Moderna Inc., Scout Bio, Passage Bio, Amicus Therapeutics, and Surmount Bio which are licensees of Penn technology. JMW is an inventor on patents that have been licensed to various biopharmaceutical companies and for which he may receive payments. C. Hinderer is an inventor on patents licensed to biopharmaceutical companies and holds equity in Scout Bio.
Figures


References
-
- Gass J, Cannon A, Mackenzie IR, et al. Mutations in progranulin are a major cause of ubiquitin‐positive frontotemporal lobar degeneration. Hum Mol Genet 2006. 15(20):2988–3001. - PubMed
-
- Gotzl JK, Mori K, Damme M, et al. Common pathobiochemical hallmarks of progranulin‐associated frontotemporal lobar degeneration and neuronal ceroid lipofuscinosis. Acta Neuropathol 2014;127(6):845–860. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
