Genome-Wide Identification and Expression Analysis of the Strawberry FvbZIP Gene Family and the Role of Key Gene FabZIP46 in Fruit Resistance to Gray Mold
- PMID: 32937812
- PMCID: PMC7569810
- DOI: 10.3390/plants9091199
Genome-Wide Identification and Expression Analysis of the Strawberry FvbZIP Gene Family and the Role of Key Gene FabZIP46 in Fruit Resistance to Gray Mold
Abstract
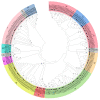
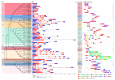
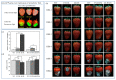
A total of 54 FvbZIP genes were identified from the strawberry genome. These genes were found to be unevenly distributed on seven different chromosomes, and two of the genes had no matching chromosomal localization. FvbZIP genes were divided into 10 subfamilies according to protein sequence, and the structures of these genes were found to be highly conserved. Based on the bioinformatics analysis of FvbZIP genes, the expression of FabZIP genes changed during different stages of its growth and of its infection with gray mold disease. FabZIP46 was substantially upregulated, and its expression remained relatively high. FabZIP46 was cloned from cultivated strawberries by homologous cloning. The results of a transient transgenic assay revealed that the damage to the fruit tissue was markedly alleviated in strawberries overexpressing FabZIP46, with the incidence rate being substantially lower than that in the control group. By contrast, a brief silencing of FabZIP46 had the opposite effect. The results revealed that FabZIP46 played a positive role in the resistance of strawberries to Botrytis cinerea. The study findings provide valuable insights into the role of bZIP transcription factors as well as a theoretical reference for the regulation of resistance to gray mold disease in strawberry fruit.
Keywords: FabZIP46; bZIP transcription factors; bioinformatics; gray mold disease; strawberry.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
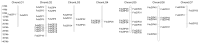


Similar articles
-
Botrytis fragariae, a New Species Causing Gray Mold on Strawberries, Shows High Frequencies of Specific and Efflux-Based Fungicide Resistance.Appl Environ Microbiol. 2017 Apr 17;83(9):e00269-17. doi: 10.1128/AEM.00269-17. Print 2017 May 1. Appl Environ Microbiol. 2017. PMID: 28235878 Free PMC article.
-
First Report of Fludioxonil Resistance in Botrytis cinerea, the Causal Agent of Gray Mold, from Strawberry Fields in Maryland and South Carolina.Plant Dis. 2014 May;98(5):692. doi: 10.1094/PDIS-08-13-0875-PDN. Plant Dis. 2014. PMID: 30708511
-
First Report of Gray Mold of Strawberry Caused by Botrytis cinerea in South Carolina.Plant Dis. 2011 Nov;95(11):1482. doi: 10.1094/PDIS-06-11-0529. Plant Dis. 2011. PMID: 30731771
-
First Report of Fludioxonil Resistance in Botrytis cinerea from a Strawberry Field in Virginia.Plant Dis. 2013 Jun;97(6):848. doi: 10.1094/PDIS-01-13-0012-PDN. Plant Dis. 2013. PMID: 30722628
-
First Report of Thiophanate-Methyl Resistance in Botrytis cinerea on Strawberry from South Carolina.Plant Dis. 2012 Nov;96(11):1700. doi: 10.1094/PDIS-06-12-0557-PDN. Plant Dis. 2012. PMID: 30727508
Cited by
-
Genomic characterization of bZIP gene family and patterns of gene regulation on Cercospora beticola Sacc resistance in sugar beet (Beta vulgaris L.).Front Genet. 2024 Jul 30;15:1430589. doi: 10.3389/fgene.2024.1430589. eCollection 2024. Front Genet. 2024. PMID: 39139817 Free PMC article.
-
Deciphering the Molecular Signatures Associated With Resistance to Botrytis cinerea in Strawberry Flower by Comparative and Dynamic Transcriptome Analysis.Front Plant Sci. 2022 May 27;13:888939. doi: 10.3389/fpls.2022.888939. eCollection 2022. Front Plant Sci. 2022. PMID: 35720571 Free PMC article.
-
RcTGA1 and glucosinolate biosynthesis pathway involvement in the defence of rose against the necrotrophic fungus Botrytis cinerea.BMC Plant Biol. 2021 May 17;21(1):223. doi: 10.1186/s12870-021-02973-z. BMC Plant Biol. 2021. PMID: 34001006 Free PMC article.
-
Strawberry FaWRKY25 Transcription Factor Negatively Regulated the Resistance of Strawberry Fruits to Botrytis cinerea.Genes (Basel). 2020 Dec 31;12(1):56. doi: 10.3390/genes12010056. Genes (Basel). 2020. PMID: 33396436 Free PMC article.
-
Insights into transcription factors controlling strawberry fruit development and ripening.Front Plant Sci. 2022 Oct 10;13:1022369. doi: 10.3389/fpls.2022.1022369. eCollection 2022. Front Plant Sci. 2022. PMID: 36299782 Free PMC article. Review.
References
-
- Amselem J., Cuomo C.A., Van Kan J.A., Viaud M., Benito E.P., Couloux A., Coutinho P.M., de Vries R.P., Dyer P.S., Fillinger S., et al. Genomic analysis of the necrotrophic fungal pathogens Sclerotinia sclerotiorum and Botrytis cinerea. PLoS Genet. 2011;7:e1002230. doi: 10.1371/journal.pgen.1002230. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

