Overcoming Resistance to Drugs Targeting KRASG12C Mutation
- PMID: 32939510
- PMCID: PMC7491749
- DOI: 10.1016/j.xinn.2020.100035
Overcoming Resistance to Drugs Targeting KRASG12C Mutation
Abstract
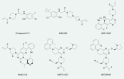
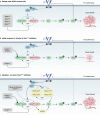
Activating KRAS mutations are present in 25% of human cancer. Although oncogenic Ras was deemed "undruggable" in the past, recent efforts led to the development of pharmacological inhibitors targeting the KRASG12C mutant, which have shown promise in early clinical trials. The development of allele-specific K-RasG12C inhibitors marked a new chapter in targeting oncogenic KRAS mutant in cancer. However, drug resistance against these new drugs will likely limit their efficacy in the clinic. Genome-wide approaches have been used to interrogate the mechanisms of resistance to K-RasG12C inhibitors, which would facilitate the development of therapeutics overcoming drug resistance. This article reviews the latest progress in resistance to K-RasG12C-targeted therapies and aims to provide insight in future research targeting drug resistance in cancer.
Keywords: DRUG RESISTANCE; KRAS INHIBITOR; ONCOGENE; TARGETED THERAPY.
Conflict of interest statement
The authors declare no competing interests.
Figures



References
-
- Bos J.L. Ras oncogenes in human cancer: a review. Cancer Res. 1989;49:4682–4689. - PubMed
-
- Patricelli M.P., Janes M.R., Li L.-S., Hansen R., Peters U., Kessler L.V., Chen Y., Kucharski J.M., Feng J., Ely T., et al. Selective inhibition of oncogenic KRAS output with small molecules targeting the inactive state. Cancer Discov. 2016;6:316–329. - PubMed
-
- Janes M.R., Zhang J., Li L.-S., Hansen R., Peters U., Guo X., Chen Y., Babbar A., Firdaus S.J., Darjania L., et al. Targeting KRAS mutant cancers with a covalent G12C-specific inhibitor. Cell. 2018;172:578–589.e17. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

