The Impact of Age on Survival in CLL Patients Receiving Ibrutinib as Initial Therapy
- PMID: 32943973
- PMCID: PMC7473982
- DOI: 10.2147/BLCTT.S262592
The Impact of Age on Survival in CLL Patients Receiving Ibrutinib as Initial Therapy
Abstract
Introduction: Recent randomized trials have demonstrated the efficacy of ibrutinib-based therapy in the treatment of patients with CLL. In Alliance A041202, a higher than expected number of unexplained deaths were reported with front-line ibrutinib in a patient population aged at least 65 years compared to ECOG 1912, which included patients up to 70 years of age.
Methods: Therefore, we conducted a retrospective analysis to investigate whether ibrutinib was associated with a greater mortality in older patients outside of a clinical trial setting. This multicenter analysis was performed by investigators at 20 academic and community practices.
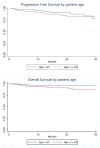
Results: Amongst the 391 patients included, there was no correlation between age and response rate, PFS, or OS. However, there was a trend to higher rate of deaths in patients >65-years-old (8.7% vs 3.8%, p=0.097), with an increased number of early deaths (13 vs 4, p=0.3).
Conclusion: These data suggest greater intolerance, and possibly mortality, with ibrutinib in an older population. Patients should be educated regarding the potential complications related to ibrutinib and symptoms of concern to report.
Keywords: CLL; elderly; ibrutinib.
© 2020 Ujjani et al.
Conflict of interest statement
Chaitra Ujjani reports non-financial supports from Pharmacyclics, Abbvie, Genentech, AstraZeneca, and Verastem. Anthony Mato reports grants and personal fees from TG Therapeutics, Celgene, Abbvie, Loxo, Sunesis, Genentech, DTRM, AstraZeneca, Octopharma, Janssen, Regeneron, Adaptive, and Pharmacyclics. Brian T Hill reports grants, personal fees from Pharmacyclics, Abbvie, AstraZeneca. John N Allan reports personal fees from Abbvie, Janssen, Pharmacyclics, Genentech, TG Therapeutics, Verastem, Sunesis, and Ascentage Pharma Group. Frederick Lansigan reports consulting fees from Acrotech and is part of the steering committee for Celgene. Alan Skarbnik is a consultant for Janssen, Pharmacyclics, Abbvie, Celgene, Kite, Alexion, Jazz Pharmaceuticals, and AstraZeneca and received payments for lectures including service on speakers bureaus from Janssen, Pharmacyclics, Abbvie, Celgene, Kite, Alexion, Jazz Pharmaceuticals, AstraZeneca, Beigene, Seattle Genetics, Genentech, and Verastem. Ryan Jacobs reports grants and/or personal fees from Pharmacyclics, Genentech, Abbvie, Verastem, TG Therapeutics, Janssen, Astra Zeneca. John Pagel reports personal fees from Gilead, Beigene, Loxo, and AstraZeneca. Danielle Brander reports grants, non-financial support from AbbVie, ArQule, Ascentage, Astra Zeneca, BeiGene, DTRM, Genentech, Juno/Celgene/BMS, MEI Pharma, Pharmacyclics, Pfizer, Teva, TG Therapeutics, Tolero, Verastem, and memberships with other guidelines/registry memberships (when sponsored or consultant also included under sponsor mentioned): NCCN panel member, informCLL registry steering committee (AbbVie), REAL registry steering committee (Verastem), and Biosimilars outcomes research panel (Pfizer). Bruce Cheson reports personal fees from Abbvie, Pharmacyclics, Celgene, TG Therapeutics, Beigene, AstraZeneca, and Genentech. Paul Barr is consultant for Abbvie, Pharmacyclics, Gilead, Genentech, TG therapeutics, Seattle Genetics, Celgene, Morphosys, Verastem, AstraZeneca, and Janssen. Lindsey Roeker reports family member with minority ownership interest in AbbVie and Abbott Laboratories. Andre Goy reports personal fees from Janssen, Pharmacyclics, during the conduct of the study; as principal investigator for and received funding for institution from Genentech-Hoffman La Roche, including personal fees from Acerta, AstraZeneca, Celgene, Constellation, Infinity, Infinity Verastem, Karyopharm, Kite pharma, Elsevier’s PracticeUpdate Oncology, Gilead, Medscape, MJH Associates, OncLive Peer Exchange, Physcians Education Resource, LLC, Xcenda, and COTA, outside the submitted work. Stephen J Schuster reports personal fees and/or grants from AstraZeneca, Merck, Novartis, Juno/Celgene, Genentech/Roche, Loxo Oncology, Tessa Therapeutics, AlloGene, BeiGene, and Celgene. Dr Nicole Lamanna received research support to institution and advisory board honorarium from Abbvie, Astra Zeneca, Beigene, Genentech, Gilead, Juno, Janssen, Mingsight, Pharmacyclics, TG Therapeutics, Celgene, and Oncternal. Constantine Tam reports personal fees from Abbvie, Janssen, Pharmacyclics. Mazyar Shadman reports personal fees from Abbvie, Genentech, Astra Zeneca, Sound Biologics, Pharmacyclics, Verastem, ADC therapeutics, Beigene, Cellectar, BMS, Mophosys and Atara Biotherapeutics, grants from Mustang Bio, Celgene, Pharmacyclics, Gilead, Genentech, Abbvie, TG therapeutics, Beigene, Astra Zeneca, Sunesis, and Beigene. The authors report no other conflicts of interest in this work.
References
-
- Hallek M, Cheson BD, Catovsky D, et al. Guidelines for the diagnosis and treatment of chronic lymphocytic leukemia: a report from the international workshop on chronic lymphocytic leukemia updating the National Cancer Institute-working group 1996 guidelines. Blood. 2008;111(12):5446–5456. doi: 10.1182/blood-2007-06-093906 - DOI - PMC - PubMed
-
- Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assoc. 1958;53(282):457–481. doi: 10.1080/01621459.1958.10501452 - DOI
-
- Anderson P, Gill R. Cox’s regression model for counting processes: a large sample study. Ann Statist. 1982;10(4):1100–1120. doi: 10.1214/aos/1176345976 - DOI
Grants and funding
LinkOut - more resources
Full Text Sources