The rise of complement in ANCA-associated vasculitis: from marginal player to target of modern therapy
- PMID: 32946609
- PMCID: PMC7670158
- DOI: 10.1111/cei.13515
The rise of complement in ANCA-associated vasculitis: from marginal player to target of modern therapy
Abstract
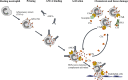
The complement system plays a central role in autoimmune diseases, including anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV). Although complement deposition is scarce in AAV pathological samples, complement activation is required for the development of necrotizing crescentic glomerulonephritis (NCGN) in mouse models of AAV and occurs via the alternative pathway. The anaphylatoxin C5a, produced by the final complement pathway, is determinant to drive the disease in animal models. C5a primes human neutrophils and enhances their activation induced by ANCA; activated neutrophils, in turn, release factors that lead to C5a generation, establishing a self-amplifying loop. C5a is also significantly increased in the serum of AAV patients with active disease compared to those in remission or healthy controls. Inhibition of the C5a receptor with avacopan is an emerging therapy that will probably allow AAV treatment with glucocorticoid-free regimens.
Keywords: ANCA; C5a; avacopan; complement; vasculitis.
© 2020 British Society for Immunology.
Conflict of interest statement
None.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

