Elaeagnus angustifolia Plant Extract Inhibits Epithelial-Mesenchymal Transition and Induces Apoptosis via HER2 Inactivation and JNK Pathway in HER2-Positive Breast Cancer Cells
- PMID: 32947764
- PMCID: PMC7570883
- DOI: 10.3390/molecules25184240
Elaeagnus angustifolia Plant Extract Inhibits Epithelial-Mesenchymal Transition and Induces Apoptosis via HER2 Inactivation and JNK Pathway in HER2-Positive Breast Cancer Cells
Abstract
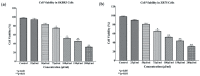
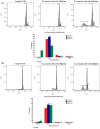
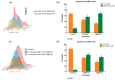
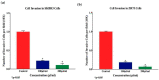
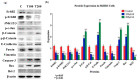
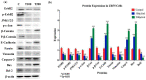
Elaeagnus angustifolia (EA) is a medicinal plant used for treating several human diseases in the Middle East. Meanwhile, the outcome of EA extract on HER2-positive breast cancer remains nascent. Thus, we herein investigated the effects of the aqueous EA extract obtained from the flowers of EA on two HER2-positive breast cancer cell lines, SKBR3 and ZR75-1. Our data revealed that EA extract inhibits cell proliferation and deregulates cell-cycle progression of these two cancer cell lines. EA extract also prevents the progression of epithelial-mesenchymal transition (EMT), an important event for cancer invasion and metastasis; this is accompanied by upregulations of E-cadherin and β-catenin, in addition to downregulations of vimentin and fascin, which are major markers of EMT. Thus, EA extract causes a drastic decrease in cell invasion ability of SKBR3 and ZR75-1 cancer cells. Additionally, we found that EA extract inhibits colony formation of both cell lines in comparison with their matched control. The molecular pathway analysis of HER2 and JNK1/2/3 of EA extract exposed cells revealed that it can block HER2 and JNK1/2/3 activities, which could be the major molecular pathway behind these events. Our findings implicate that EA extract may possess chemo-preventive effects against HER2-positive breast cancer via HER2 inactivation and specifically JNK1/2/3 signaling pathways.
Keywords: EMT; Elaeagnus angustifolia; apoptosis; breast cancer; chemoprevention.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures


Similar articles
-
Elaeagnus angustifolia Plant Extract Inhibits Angiogenesis and Downgrades Cell Invasion of Human Oral Cancer Cells via Erk1/Erk2 Inactivation.Nutr Cancer. 2018 Feb-Mar;70(2):297-305. doi: 10.1080/01635581.2018.1412472. Epub 2018 Jan 4. Nutr Cancer. 2018. PMID: 29300111
-
Novel Nitrogen-Based Chalcone Analogs Provoke Substantial Apoptosis in HER2-Positive Human Breast Cancer Cells via JNK and ERK1/ERK2 Signaling Pathways.Int J Mol Sci. 2021 Sep 6;22(17):9621. doi: 10.3390/ijms22179621. Int J Mol Sci. 2021. PMID: 34502529 Free PMC article.
-
6 α-Hydroxy-4[14], 10[15]-guainadien-8β, 12-olide induced cell cycle arrest via modulation of EMT and Wnt/β-catenin pathway in HER-2 positive breast cancer cells.J Steroid Biochem Mol Biol. 2020 Mar;197:105514. doi: 10.1016/j.jsbmb.2019.105514. Epub 2019 Oct 31. J Steroid Biochem Mol Biol. 2020. PMID: 31678110
-
Dietary plants for oral cancer prevention and therapy: A review of preclinical and clinical studies.Phytother Res. 2024 Nov;38(11):5225-5263. doi: 10.1002/ptr.8293. Epub 2024 Aug 28. Phytother Res. 2024. PMID: 39193857 Review.
-
Exploring the Potential of Ellagic Acid in Gastrointestinal Cancer Prevention: Recent Advances and Future Directions.Oncol Ther. 2024 Dec;12(4):685-699. doi: 10.1007/s40487-024-00296-1. Epub 2024 Sep 2. Oncol Ther. 2024. PMID: 39222186 Free PMC article. Review.
Cited by
-
Fascin-1: Updated biological functions and therapeutic implications in cancer biology.BBA Adv. 2022 May 17;2:100052. doi: 10.1016/j.bbadva.2022.100052. eCollection 2022. BBA Adv. 2022. PMID: 37082587 Free PMC article. Review.
-
Targeting HER2-positive breast cancer cells by a combination of dasatinib and BMS-202: Insight into the molecular pathways.Cancer Cell Int. 2024 Mar 2;24(1):94. doi: 10.1186/s12935-023-03195-z. Cancer Cell Int. 2024. PMID: 38431613 Free PMC article.
-
Biochemical and antioxidant activity of wild edible fruits of the eastern Himalaya, India.Front Nutr. 2023 Mar 1;10:1039965. doi: 10.3389/fnut.2023.1039965. eCollection 2023. Front Nutr. 2023. PMID: 36937364 Free PMC article.
-
Green synthesis of nanohydroxyapatite trough Elaeagnus angustifolia L. extract and evaluating its anti-tumor properties in MCF7 breast cancer cell line.BMC Complement Med Ther. 2023 Sep 26;23(1):338. doi: 10.1186/s12906-023-04116-3. BMC Complement Med Ther. 2023. PMID: 37752472 Free PMC article.
-
Extraction of Bound Polyphenols from Elaeagnus angustifolia L. by Ultrasonic-Assisted Enzymatic Hydrolysis and Evaluation of Its Antioxidant Activity In Vitro.Foods. 2025 Apr 29;14(9):1567. doi: 10.3390/foods14091567. Foods. 2025. PMID: 40361648 Free PMC article.
References
-
- Sareyeldin R.M., Gupta I., Al-Hashimi I., Al-Thawadi H.A., Al Farsi H.F., Vranic S., Al Moustafa A.-E. Gene Expression and miRNAs Profiling: Function and Regulation in Human Epidermal Growth Factor Receptor 2 (HER2)-Positive Breast Cancer. Cancers. 2019;11:646. doi: 10.3390/cancers11050646. - DOI - PMC - PubMed
-
- Giordano S.H., Temin S., Chandarlapaty S., Crews J.R., Esteva F.J., Kirshner J.J., Krop I.E., Levinson J., Lin N.U., Modi S., et al. Systemic Therapy for Patients with Advanced Human Epidermal Growth Factor Receptor 2–Positive Breast Cancer: ASCO Clinical Practice Guideline Update. J. Clin. Oncol. 2018;36:2736–2740. doi: 10.1200/JCO.2018.79.2697. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

