GRIP1 regulates synaptic plasticity and learning and memory
- PMID: 32948689
- PMCID: PMC7547244
- DOI: 10.1073/pnas.2014827117
GRIP1 regulates synaptic plasticity and learning and memory
Abstract
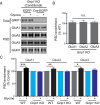
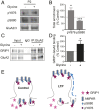
Hebbian plasticity is a key mechanism for higher brain functions, such as learning and memory. This form of synaptic plasticity primarily involves the regulation of synaptic α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor (AMPAR) abundance and properties, whereby AMPARs are inserted into synapses during long-term potentiation (LTP) or removed during long-term depression (LTD). The molecular mechanisms underlying AMPAR trafficking remain elusive, however. Here we show that glutamate receptor interacting protein 1 (GRIP1), an AMPAR-binding protein shown to regulate the trafficking and synaptic targeting of AMPARs, is required for LTP and learning and memory. GRIP1 is recruited into synapses during LTP, and deletion of Grip1 in neurons blocks synaptic AMPAR accumulation induced by glycine-mediated depolarization. In addition, Grip1 knockout mice exhibit impaired hippocampal LTP, as well as deficits in learning and memory. Mechanistically, we find that phosphorylation of serine-880 of the GluA2 AMPAR subunit (GluA2-S880) is decreased while phosphorylation of tyrosine-876 on GluA2 (GluA2-Y876) is elevated during chemically induced LTP. This enhances the strength of the GRIP1-AMPAR association and, subsequently, the insertion of AMPARs into the postsynaptic membrane. Together, these results demonstrate an essential role of GRIP1 in regulating AMPAR trafficking during synaptic plasticity and learning and memory.
Keywords: AMPA receptor; GRIP1; LTP; learning and memory; synaptic plasticity.
Conflict of interest statement
The authors declare no competing interest.
Figures



References
-
- Volk L., Chiu S. L., Sharma K., Huganir R. L., Glutamate synapses in human cognitive disorders. Annu. Rev. Neurosci. 38, 127–149 (2015). - PubMed
-
- Martin S. J., Grimwood P. D., Morris R. G., Synaptic plasticity and memory: An evaluation of the hypothesis. Annu. Rev. Neurosci. 23, 649–711 (2000). - PubMed
-
- Mansvelder H. D., Verhoog M. B., Goriounova N. A., Synaptic plasticity in human cortical circuits: Cellular mechanisms of learning and memory in the human brain? Curr. Opin. Neurobiol. 54, 186–193 (2019). - PubMed
-
- Malenka R. C., Synaptic plasticity in the hippocampus: LTP and LTD. Cell 78, 535–538 (1994). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

