Prognostic values of SNAI family members in breast cancer patients
- PMID: 32953722
- PMCID: PMC7475426
- DOI: 10.21037/atm-20-681
Prognostic values of SNAI family members in breast cancer patients
Abstract
Background: Breast cancer (BC) is one of the most lethal malignant tumors and the leading cause of cancer-related death worldwide. Although early diagnostic techniques for BC have been well developed, 40% of cases are still diagnosed at the advanced stage, while for BC patients with distant metastases, the 5-year survival rate is usually lower than 30%. The Snail family, generally regarded as transcriptional repressors, has been indicated to be an essential prognostic factor in malignant tumors. However, limited data exist on public databases concerning the prognostic value of individual Snail family members in BC, especially SNAI3.
Methods: Data from public databases including cBioPortal for Cancer Genomics, Gene Expression Omnibus, UCSC Xena Browser, and Human Protein Atlas (HPA) were downloaded. Based on the Kaplan¬-Meier plotter platform, correlation of the three members of the Snail family and prognosis in BC were analyzed. Individual Snail family members and their co-expressed genes were respectively enriched on different pathways and biological processes via the functional enrichment analysis (FunRich) tool.
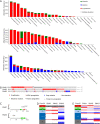
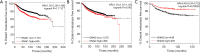
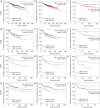
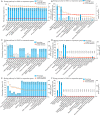
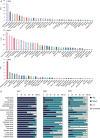
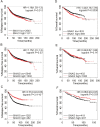
Results: High SNAI1 mRNA expression was associated with shorter distant metastasis-free survival (DMFS) in all BC patients regardless of PAM50 subtype. Conversely, high SNAI3 mRNA expression was associated with longer DMFS. Although the presence of SNAI2 expression was significantly associated with DMFS in the whole cohort, no significant correlation was found in patients with luminal A or HER2 subtype. For patients with the most diverse clinicopathological features, high SNAI1 expression was associated with poor survival, with the converse being true for SNAI3. However, the impact on prognosis of patients with different clinicopathological features produced by SNAI2 expression was inconclusive. Furthermore, we discovered that SNAI1 or SNAI2 and their co-expressed genes frequently enriched receptor tyrosine kinase (RTK) signaling and integrin-related pathways which mainly functioned on epithelial-mesenchymal transition and were further involved in several processes of signal transduction and cell communication. Furthermore, as SNAI3, along with its co-expressed genes, enriched immune-related pathways, it may thus play a role in mediating the immune system.
Conclusions: Our analysis revealed that SNAI1 mRNA expression may potentially be a negative prognostic factor, whereas SNAI3 mRNA was associated with positive prognosis in BC. Therefore, the assessment of SNAI1 and SNAI3 expression may be valuable for predicting prognosis in BC patients.
Keywords: Breast cancer (BC); SNAI family; mRNA; prognosis.
2020 Annals of Translational Medicine. All rights reserved.
Conflict of interest statement
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/atm-20-681). The authors have no conflicts of interest to declare.
Figures
Similar articles
-
Comparative genomics on SNAI1, SNAI2, and SNAI3 orthologs.Oncol Rep. 2005 Oct;14(4):1083-6. Oncol Rep. 2005. PMID: 16142376
-
Analysis of the Effect of SNAI Family in Breast Cancer and Immune Cell.Front Cell Dev Biol. 2022 Jul 8;10:906885. doi: 10.3389/fcell.2022.906885. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35898399 Free PMC article.
-
Identification and characterization of human SNAIL3 (SNAI3) gene in silico.Int J Mol Med. 2003 Mar;11(3):383-8. Int J Mol Med. 2003. PMID: 12579345
-
Expressions and prognostic values of the E2F transcription factors in human breast carcinoma.Cancer Manag Res. 2018 Sep 13;10:3521-3532. doi: 10.2147/CMAR.S172332. eCollection 2018. Cancer Manag Res. 2018. PMID: 30271201 Free PMC article. Review.
-
The Value of EphB2 Receptor and Cognate Ephrin Ligands in Prognostic and Predictive Assessments of Human Breast Cancer.Int J Mol Sci. 2021 Jul 28;22(15):8098. doi: 10.3390/ijms22158098. Int J Mol Sci. 2021. PMID: 34360867 Free PMC article.
Cited by
-
Predictive significance of HIF-1α, Snail, and PD-L1 expression in breast cancer.Clin Exp Med. 2023 Oct;23(6):2369-2383. doi: 10.1007/s10238-023-01026-z. Epub 2023 Feb 21. Clin Exp Med. 2023. PMID: 36802309
-
Prognostic and clinicopathological value of Slug protein expression in breast cancer: a systematic review and meta-analysis.World J Surg Oncol. 2022 Nov 14;20(1):361. doi: 10.1186/s12957-022-02825-6. World J Surg Oncol. 2022. PMID: 36372891 Free PMC article.
-
Prognostic and Predictive Value of Transcription Factors Panel for Digestive System Carcinoma.Front Oncol. 2021 Oct 21;11:670129. doi: 10.3389/fonc.2021.670129. eCollection 2021. Front Oncol. 2021. PMID: 34745933 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
