CERE-120 Prevents Irradiation-Induced Hypofunction and Restores Immune Homeostasis in Porcine Salivary Glands
- PMID: 32953934
- PMCID: PMC7479444
- DOI: 10.1016/j.omtm.2020.07.016
CERE-120 Prevents Irradiation-Induced Hypofunction and Restores Immune Homeostasis in Porcine Salivary Glands
Abstract
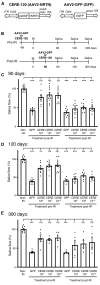
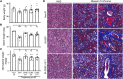
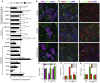
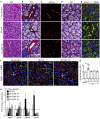
Salivary gland hypofunction causes significant morbidity and loss of quality of life for head and neck cancer patients treated with radiotherapy. Preventing hypofunction is an unmet therapeutic need. We used an adeno-associated virus serotype 2 (AAV2) vector expressing the human neurotrophic factor neurturin (CERE-120) to treat murine submandibular glands either pre- or post-irradiation (IR). Treatment with CERE-120 pre-IR, not post-IR, prevented hypofunction. RNA sequencing (RNA-seq) analysis showed reduced gene expression associated with fibrosis and the innate and humoral immune responses. We then used a minipig model with CERE-120 treatment pre-IR and also compared outcomes of the contralateral non-IR gland. Analysis of gene expression, morphology, and immunostaining showed reduced IR-related immune responses and improved secretory mechanisms. CERE-120 prevented IR-induced hypofunction and restored immune homeostasis, and there was a coordinated contralateral gland response to either damage or treatment. CERE-120 gene therapy is a potential treatment for head and neck cancer patients to influence communication among neuronal, immune, and epithelial cells to prevent IR-induced salivary hypofunction and restore immune homeostasis.
Keywords: AAV-neurturin; CERE-120; fibrosis; gene therapy; head and neck cancer; humoral immunity; innate immunity; irradiation damage; salivary gland; salivary hypofunction.
Figures


Similar articles
-
Neurturin Gene Therapy Protects Parasympathetic Function to Prevent Irradiation-Induced Murine Salivary Gland Hypofunction.Mol Ther Methods Clin Dev. 2018 Feb 23;9:172-180. doi: 10.1016/j.omtm.2018.02.008. eCollection 2018 Jun 15. Mol Ther Methods Clin Dev. 2018. PMID: 29560384 Free PMC article.
-
Prevention of radiation-induced salivary hypofunction following hKGF gene delivery to murine submandibular glands.Clin Cancer Res. 2011 May 1;17(9):2842-51. doi: 10.1158/1078-0432.CCR-10-2982. Epub 2011 Mar 2. Clin Cancer Res. 2011. PMID: 21367751 Free PMC article.
-
Hypoxia-Activated Adipose Mesenchymal Stem Cells Prevents Irradiation-Induced Salivary Hypofunction by Enhanced Paracrine Effect Through Fibroblast Growth Factor 10.Stem Cells. 2018 Jul;36(7):1020-1032. doi: 10.1002/stem.2818. Epub 2018 Apr 10. Stem Cells. 2018. PMID: 29569790
-
Advancing neurotrophic factors as treatments for age-related neurodegenerative diseases: developing and demonstrating "clinical proof-of-concept" for AAV-neurturin (CERE-120) in Parkinson's disease.Neurobiol Aging. 2013 Jan;34(1):35-61. doi: 10.1016/j.neurobiolaging.2012.07.018. Epub 2012 Aug 24. Neurobiol Aging. 2013. PMID: 22926166 Review.
-
Application of regenerative medicine to salivary gland hypofunction.Jpn Dent Sci Rev. 2021 Nov;57:54-59. doi: 10.1016/j.jdsr.2021.03.002. Epub 2021 Apr 27. Jpn Dent Sci Rev. 2021. PMID: 33995711 Free PMC article. Review.
Cited by
-
Indomethacin Treatment Post-irradiation Improves Mouse Parotid Salivary Gland Function via Modulation of Prostaglandin E2 Signaling.Front Bioeng Biotechnol. 2021 Jul 21;9:697671. doi: 10.3389/fbioe.2021.697671. eCollection 2021. Front Bioeng Biotechnol. 2021. PMID: 34381764 Free PMC article.
-
Neurotrophin signaling is a central mechanism of salivary dysfunction after irradiation that disrupts myoepithelial cells.NPJ Regen Med. 2023 Mar 25;8(1):17. doi: 10.1038/s41536-023-00290-7. NPJ Regen Med. 2023. PMID: 36966175 Free PMC article.
-
Matrix Degradability Contributes to the Development of Salivary Gland Progenitor Cells with Secretory Functions.ACS Appl Mater Interfaces. 2023 Jul 12;15(27):32148-32161. doi: 10.1021/acsami.3c03064. Epub 2023 Jun 26. ACS Appl Mater Interfaces. 2023. PMID: 37364369 Free PMC article.
-
Autologous mesenchymal stem cells offer a new paradigm for salivary gland regeneration.Int J Oral Sci. 2023 May 10;15(1):18. doi: 10.1038/s41368-023-00224-5. Int J Oral Sci. 2023. PMID: 37165024 Free PMC article. Review.
-
Bringing hydrogel-based craniofacial therapies to the clinic.Acta Biomater. 2022 Jan 15;138:1-20. doi: 10.1016/j.actbio.2021.10.056. Epub 2021 Nov 4. Acta Biomater. 2022. PMID: 34743044 Free PMC article. Review.
References
-
- Vissink A., Mitchell J.B., Baum B.J., Limesand K.H., Jensen S.B., Fox P.C., Elting L.S., Langendijk J.A., Coppes R.P., Reyland M.E. Clinical management of salivary gland hypofunction and xerostomia in head-and-neck cancer patients: successes and barriers. Int. J. Radiat. Oncol. Biol. Phys. 2010;78:983–991. - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

