Dry Two-Step Self-Assembly of Stable Supported Lipid Bilayers on Silicon Substrates
- PMID: 32957654
- PMCID: PMC7555443
- DOI: 10.3390/ijms21186819
Dry Two-Step Self-Assembly of Stable Supported Lipid Bilayers on Silicon Substrates
Abstract
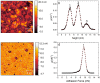
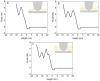
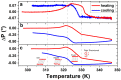
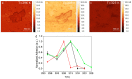
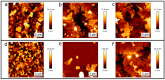
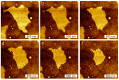
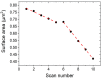
Artificial membranes are models for biological systems and are important for applications. We introduce a dry two-step self-assembly method consisting of the high-vacuum evaporation of phospholipid molecules over silicon, followed by a subsequent annealing step in air. We evaporate dipalmitoylphosphatidylcholine (DPPC) molecules over bare silicon without the use of polymer cushions or solvents. High-resolution ellipsometry and AFM temperature-dependent measurements are performed in air to detect the characteristic phase transitions of DPPC bilayers. Complementary AFM force-spectroscopy breakthrough events are induced to detect single- and multi-bilayer formation. These combined experimental methods confirm the formation of stable non-hydrated supported lipid bilayers with phase transitions gel to ripple at 311.5 ± 0.9 K, ripple to liquid crystalline at 323.8 ± 2.5 K and liquid crystalline to fluid disordered at 330.4 ± 0.9 K, consistent with such structures reported in wet environments. We find that the AFM tip induces a restructuring or intercalation of the bilayer that is strongly related to the applied tip-force. These dry supported lipid bilayers show long-term stability. These findings are relevant for the development of functional biointerfaces, specifically for fabrication of biosensors and membrane protein platforms. The observed stability is relevant in the context of lifetimes of systems protected by bilayers in dry environments.
Keywords: artificial membranes; atomic force microscopy; bio-silica interfaces; force spectroscopy; high resolution ellipsometry; phase transitions; physical vapor deposition; self-assembly; supported lipid bilayers.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
Similar articles
-
Towards bio-silicon interfaces: formation of an ultra-thin self-hydrated artificial membrane composed of dipalmitoylphosphatidylcholine (DPPC) and chitosan deposited in high vacuum from the gas-phase.J Chem Phys. 2014 Sep 14;141(10):104201. doi: 10.1063/1.4894224. J Chem Phys. 2014. PMID: 25217908
-
Temperature-controlled structure and kinetics of ripple phases in one- and two-component supported lipid bilayers.Biophys J. 2003 Jul;85(1):350-60. doi: 10.1016/S0006-3495(03)74479-8. Biophys J. 2003. PMID: 12829489 Free PMC article.
-
Direct submolecular scale imaging of mesoscale molecular order in supported dipalmitoylphosphatidylcholine bilayers.Langmuir. 2011 Apr 5;27(7):3749-53. doi: 10.1021/la104640v. Epub 2011 Mar 3. Langmuir. 2011. PMID: 21370902
-
Phase transitions in supported lipid bilayers studied by AFM.Soft Matter. 2014 Oct 7;10(37):7145-64. doi: 10.1039/c4sm01104j. Soft Matter. 2014. PMID: 25090108 Review.
-
Force spectroscopy as a tool to investigate the properties of supported lipid membranes.Microsc Res Tech. 2010 Oct;73(10):965-72. doi: 10.1002/jemt.20834. Microsc Res Tech. 2010. PMID: 20232466 Review.
References
-
- Kustanovich K., Yantchev V., Doosti B.A., Gözen I., Jesorka A. A microfluidics-integrated impedance/surface acoustic resonance tandem sensor. Sens. Bio-Sens. Res. 2019;25:100291. doi: 10.1016/j.sbsr.2019.100291. - DOI
-
- Czolkos I., Jesorka A., Orwar O. Molecular phospholipid films on solid supports. Soft Matter. 2011;7:4562–4576. doi: 10.1039/c0sm01212b. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

