Unique expansion of IL-21+ Tfh and Tph cells under control of ICOS identifies Sjögren's syndrome with ectopic germinal centres and MALT lymphoma
- PMID: 32963045
- PMCID: PMC7677495
- DOI: 10.1136/annrheumdis-2020-217646
Unique expansion of IL-21+ Tfh and Tph cells under control of ICOS identifies Sjögren's syndrome with ectopic germinal centres and MALT lymphoma
Abstract
Objectives: To explore the relevance of T-follicular-helper (Tfh) and pathogenic peripheral-helper T-cells (Tph) in promoting ectopic lymphoid structures (ELS) and B-cell mucosa-associated lymphoid tissue (MALT) lymphomas (MALT-L) in Sjögren's syndrome (SS) patients.
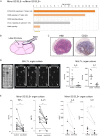
Methods: Salivary gland (SG) biopsies with matched peripheral blood were collected from four centres across the European Union. Transcriptomic (microarray and quantitative PCR) analysis, FACS T-cell immunophenotyping with intracellular cytokine detection, multicolor immune-fluorescence microscopy and in situ hybridisation were performed to characterise lesional and circulating Tfh and Tph-cells. SG-organ cultures were used to investigate functionally the blockade of T-cell costimulatory pathways on key proinflammatory cytokine production.
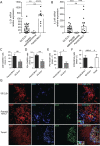
Results: Transcriptomic analysis in SG identified Tfh-signature, interleukin-21 (IL-21) and the inducible T-cell co-stimulator (ICOS) costimulatory pathway as the most upregulated genes in ELS+SS patients, with parotid MALT-L displaying a 400-folds increase in IL-21 mRNA. Peripheral CD4+CXC-motif chemokine receptor 5 (CXCR5)+programmed cell death protein 1 (PD1)+ICOS+ Tfh-like cells were significantly expanded in ELS+SS patients, were the main producers of IL-21, and closely correlated with circulating IgG and reduced complement C4. In the SG, lesional CD4+CD45RO+ICOS+PD1+ cells selectively infiltrated ELS+ tissues and were aberrantly expanded in parotid MALT-L. In ELS+SG and MALT-L parotids, conventional CXCR5+CD4+PD1+ICOS+Foxp3- Tfh-cells and a uniquely expanded population of CXCR5-CD4+PD1hiICOS+Foxp3- Tph-cells displayed frequent IL-21/interferon-γ double-production but poor IL-17 expression. Finally, ICOS blockade in ex vivo SG-organ cultures significantly reduced the production of IL-21 and inflammatory cytokines IL-6, IL-8 and tumour necrosis factor-α (TNF-α).
Conclusions: Overall, these findings highlight Tfh and Tph-cells, IL-21 and the ICOS costimulatory pathway as key pathogenic players in SS immunopathology and exploitable therapeutic targets in SS.
Keywords: autoimmune diseases; cytokines; sjogren's syndrome; t-lymphocyte subsets.
© Author(s) (or their employer(s)) 2020. Re-use permitted under CC BY. Published by BMJ.
Conflict of interest statement
Competing interests: BF: Consultancy for Novartis, Roche, and BMS. SJB: Consultancy for Astrazeneca, Biogen, BMS, Celgene, Medimmune, MTPharma, Novartis, Ono, UCB, xtlbio. MB: consultancy and/or unrestricted grant support from Medimmune, GSK, Janssen, UCB. GC and JC are AstraZeneca employees and own company stocks.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

