From statistical regularities in multisensory inputs to peripersonal space representation and body ownership: Insights from a neural network model
- PMID: 32965729
- PMCID: PMC7894138
- DOI: 10.1111/ejn.14981
From statistical regularities in multisensory inputs to peripersonal space representation and body ownership: Insights from a neural network model
Abstract
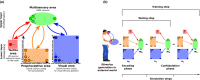
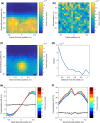
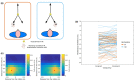
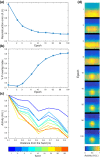
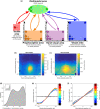
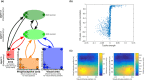
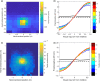
Peripersonal space (PPS), the interface between the self and the environment, is represented by a network of multisensory neurons with visual (or auditory) receptive fields anchored to specific body parts, and tactile receptive fields covering the same body parts. Neurophysiological and behavioural features of hand PPS representation have been previously modelled through a neural network constituted by one multisensory population integrating tactile inputs with visual/auditory external stimuli. Reference frame transformations were not explicitly modelled, as stimuli were encoded in pre-computed hand-centred coordinates. Here we present a novel model, aiming to overcome this limitation by including a proprioceptive population encoding hand position. We confirmed behaviourally the plausibility of the proposed architecture, showing that visuo-proprioceptive information is integrated to enhance tactile processing on the hand. Moreover, the network's connectivity was spontaneously tuned through a Hebbian-like mechanism, under two minimal assumptions. First, the plasticity rule was designed to learn the statistical regularities of visual, proprioceptive and tactile inputs. Second, such statistical regularities were simply those imposed by the body structure. The network learned to integrate proprioceptive and visual stimuli, and to compute their hand-centred coordinates to predict tactile stimulation. Through the same mechanism, the network reproduced behavioural correlates of manipulations implicated in subjective body ownership: the invisible and the rubber hand illusion. We thus propose that PPS representation and body ownership may emerge through a unified neurocomputational process; the integration of multisensory information consistently with a model of the body in the environment, learned from the natural statistics of sensory inputs.
Keywords: Hebbian learning; bodily self-consciousness; body representation; reference frame transformations; statistical inference.
© 2020 The Authors. European Journal of Neuroscience published by Federation of European Neuroscience Societies and John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
The magnetic touch illusion: A perceptual correlate of visuo-tactile integration in peripersonal space.Cognition. 2016 Oct;155:44-56. doi: 10.1016/j.cognition.2016.06.004. Epub 2016 Jun 24. Cognition. 2016. PMID: 27348406
-
Unconscious integration of multisensory bodily inputs in the peripersonal space shapes bodily self-consciousness.Cognition. 2017 Sep;166:174-183. doi: 10.1016/j.cognition.2017.05.028. Epub 2017 Jun 1. Cognition. 2017. PMID: 28577447
-
Sleep dissolves illusion: sleep withstands learning of visuo-tactile-proprioceptive integration induced by repeated days of rubber hand illusion training.PLoS One. 2014 Jan 17;9(1):e85734. doi: 10.1371/journal.pone.0085734. eCollection 2014. PLoS One. 2014. PMID: 24465671 Free PMC article. Clinical Trial.
-
Peripersonal space (PPS) as a multisensory interface between the individual and the environment, defining the space of the self.Neurosci Biobehav Rev. 2019 Apr;99:138-159. doi: 10.1016/j.neubiorev.2019.01.016. Epub 2019 Jan 24. Neurosci Biobehav Rev. 2019. PMID: 30685486 Review.
-
From multisensory integration in peripersonal space to bodily self-consciousness: from statistical regularities to statistical inference.Ann N Y Acad Sci. 2018 Jun 6. doi: 10.1111/nyas.13867. Online ahead of print. Ann N Y Acad Sci. 2018. PMID: 29876922 Review.
Cited by
-
Grounding Context in Embodied Cognitive Robotics.Front Neurorobot. 2022 Jun 15;16:843108. doi: 10.3389/fnbot.2022.843108. eCollection 2022. Front Neurorobot. 2022. PMID: 35812785 Free PMC article.
-
The multisensory mind: a systematic review of multisensory integration processing in Anorexia and Bulimia Nervosa.J Eat Disord. 2023 Nov 16;11(1):204. doi: 10.1186/s40337-023-00930-9. J Eat Disord. 2023. PMID: 37974266 Free PMC article. Review.
-
Visual stimuli in the peripersonal space facilitate the spatial prediction of tactile events-A comparison between approach and nearness effects.Front Hum Neurosci. 2023 Oct 12;17:1203100. doi: 10.3389/fnhum.2023.1203100. eCollection 2023. Front Hum Neurosci. 2023. PMID: 37900729 Free PMC article.
-
Spatial proximity to others induces plastic changes in the neural representation of the peripersonal space.iScience. 2022 Dec 26;26(1):105879. doi: 10.1016/j.isci.2022.105879. eCollection 2023 Jan 20. iScience. 2022. PMID: 36654859 Free PMC article.
-
Peripersonal space: why so last-second?Philos Trans R Soc Lond B Biol Sci. 2024 Oct 7;379(1911):20230159. doi: 10.1098/rstb.2023.0159. Epub 2024 Aug 19. Philos Trans R Soc Lond B Biol Sci. 2024. PMID: 39155714 Free PMC article. Review.
References
-
- Alais, D. , & Burr, D. (2004). The ventriloquist effect results from near‐optimal bimodal integration. Current Biology, 14, 257–262. - PubMed
-
- Avillac, M. , Denève, S. , Olivier, E. , Pouget, A. , & Duhamel, J.‐R. (2005). Reference frames for representing visual and tactile locations in parietal cortex. Nature Neuroscience, 8, 941–949. - PubMed
-
- Barlow, H. B. (1961). Possible principles underlying the transformation of sensory messages. Sensory Communication, 217–234.
-
- Bernasconi, F. , Noel, J. , Park, H. D. , Faivre, N. , Seeck, M. , Spinelli, L. , Schaller, K. , Blanke, O. , & Serino, A. (2018). Audio‐tactile and peripersonal space processing around the trunk in human parietal and temporal cortex: An intracranial EEG study. Cerebral Cortex, 28, 3385–3397. 10.1093/cercor/bhy156 - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

