Factors associated with outcomes after a second CD19-targeted CAR T-cell infusion for refractory B-cell malignancies
- PMID: 32967009
- PMCID: PMC7819764
- DOI: 10.1182/blood.2020006770
Factors associated with outcomes after a second CD19-targeted CAR T-cell infusion for refractory B-cell malignancies
Abstract
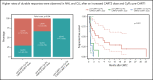
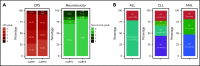
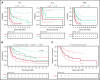
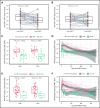
CD19-targeted chimeric antigen receptor-engineered (CD19 CAR) T-cell therapy has shown significant efficacy for relapsed or refractory (R/R) B-cell malignancies. Yet, CD19 CAR T cells fail to induce durable responses in most patients. Second infusions of CD19 CAR T cells (CART2) have been considered as a possible approach to improve outcomes. We analyzed data from 44 patients with R/R B-cell malignancies (acute lymphoblastic leukemia [ALL], n = 14; chronic lymphocytic leukemia [CLL], n = 9; non-Hodgkin lymphoma [NHL], n = 21) who received CART2 on a phase 1/2 trial (NCT01865617) at our institution. Despite a CART2 dose increase in 82% of patients, we observed a low incidence of severe toxicity after CART2 (grade ≥3 cytokine release syndrome, 9%; grade ≥3 neurotoxicity, 11%). After CART2, complete response (CR) was achieved in 22% of CLL, 19% of NHL, and 21% of ALL patients. The median durations of response after CART2 in CLL, NHL, and ALL patients were 33, 6, and 4 months, respectively. Addition of fludarabine to cyclophosphamide-based lymphodepletion before the first CAR T-cell infusion (CART1) and an increase in the CART2 dose compared with CART1 were independently associated with higher overall response rates and longer progression-free survival after CART2. We observed durable CAR T-cell persistence after CART2 in patients who received cyclophosphamide and fludarabine (Cy-Flu) lymphodepletion before CART1 and a higher CART2 compared with CART1 cell dose. The identification of 2 modifiable pretreatment factors independently associated with better outcomes after CART2 suggests strategies to improve in vivo CAR T-cell kinetics and responses after repeat CAR T-cell infusions, and has implications for the design of trials of novel CAR T-cell products after failure of prior CAR T-cell immunotherapies.
© 2021 by The American Society of Hematology.
Conflict of interest statement
Conflict-of-interest disclosure: C.K.C. has pending patients licensed with Nektar Therapeutics. R.D.C. has received research funding from Amgen, Kite/Gilead, Merck, Pfizer, and Vanda Pharmaceuticals; honoraria/consulting from Amgen and Pfizer; and his spouse is employed by and owns stock in Seattle Genetics. A.J.C. receives research funding from Janssen, BMS, and AbbVie and is a member of scientific advisory boards for Sanofi and Cellectar; he has received consultancy honorarium from Janssen. D.J.G. has received research funding, has served as an advisor, and has received royalties from Juno Therapeutics, a Bristol-Myers Squibb company; has served as an advisor and received research funding from Seattle Genetics; has served as an advisor to GlaxoSmithKline, Janssen Biotech, and Legend Biotech; and has received research funding from Sanofi and Cellectar Biosciences. M.S. received research funding from Mustang Bio, Celgene, Pharmacyclics, Gilead, Genentech, Abbvie, TG therapeutics, Beigene, Acerta Pharma, Merck, and has served on advisory boards for Abbvie, Genentech, Astra Zeneca, Sound Biologics, Verastem, ADC therapeutics, and Atara Biotherapeutics. S.R.R. holds equity, has served as an advisor, and has patents licensed to Juno Therapeutics, a Celgene company; is a founder of Lyell Immunopharma, and has served on advisory boards for Adaptive Biotechnologies and Nohla. D.G.M. has received research funding from Kite Pharma, Juno Therapeutics, a Celgene company, and Celgene; has received honoraria for participation in advisory boards meetings with Kite Pharma, Gilead, Genentech, Novartis, and Eureka. C.J.T. receives research funding from Juno Therapeutics, Nektar Therapeutics, Minerva, TCR2, and AstraZeneca; is a member of scientific advisory boards and has options in Precision Biosciences, Eureka Therapeutics, Caribou Biosciences, Myeloid Therapeutics, and ArsenalBio; serves on scientific advisory boards for T-CURX and Century Therapeutics; has served on advisory boards for Nektar Therapeutics, Allogene, Kite/Gilead, Novartis, Humanigen, PACT Pharma, Amgen, and Astra Zeneca; and has patents licensed to Juno Therapeutics. The remaining authors declare no competing financial interests.
Figures
Comment in
-
A second CD19 CAR T-cell infusion: yes or no?Blood. 2021 Jan 21;137(3):284-286. doi: 10.1182/blood.2020009206. Blood. 2021. PMID: 33475747 No abstract available.
References
-
- Shah BD, Bishop M, Oluwole OO, et al. . End of phase I results of ZUMA-3, a phase 1/2 study of KTE-X19, anti-CD19 chimeric antigen receptor (CAR) T cell therapy, in adult patients (pts) with relapsed/refractory (R/R) acute lymphoblastic leukemia (ALL). J Clin Oncol. 2019;37(15_suppl). Abstract 7006.
-
- Grupp SA, Maude SL, Shaw PA, Aplenc R, Barrett DM. Durable remissions in children with relapsed/refractory ALL treated with T cells engineered with a CD19-targeted chimeric antigen receptor (CTL019) [abstract].Blood. 2015;126(23):681 Abstract 614.
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

