Replicate Aptima Assay for Quantifying Residual Plasma Viremia in Individuals on Antiretroviral Therapy
- PMID: 32967900
- PMCID: PMC7685884
- DOI: 10.1128/JCM.01400-20
Replicate Aptima Assay for Quantifying Residual Plasma Viremia in Individuals on Antiretroviral Therapy
Abstract
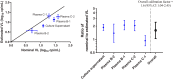
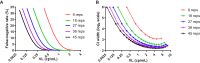
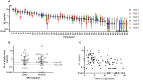
Detection of residual plasma viremia in antiretroviral therapy (ART)-suppressed HIV-infected individuals is critical for characterizing the latent reservoir and evaluating the impact of cure interventions. Ultracentrifugation-based single-copy assays are sensitive but labor intensive. Fully automated replicate testing using a standard clinical viral load assay was evaluated as a high-throughput alternative for the quantification of low-level viremia. Four plasma samples from blood donors with acute HIV-1 infection and one viral culture supernatant were serially diluted into 25-ml samples to nominal viral loads ranging from 39 to <0.5 copies (cp)/ml. Each dilution was tested with 45 replicates (reps) using 0.5 ml/rep with the Aptima HIV-1 Quant assay. The nominal and estimated viral loads based on the single-hit Poisson model were compared, and a hybrid Poisson digital model for calibrated viral load estimation was derived. Testing performed using 45 reps on longitudinal plasma samples from 50 ART-suppressed individuals in the Reservoir Assay Validation and Evaluation Network (RAVEN) study cohort (range of 1 to 19 years of continuous ART suppression) showed a median viral load of 0.54 cp/ml (interquartile range [IQR], 0.22 to 1.46 cp/ml) and a 14% (95% confidence interval [CI], 9% to 19%) decline in viral load for each additional year in duration suppressed. Within the RAVEN cohort, the expected false-negative rate for detection at lower rep numbers using 9 and 18 reps was 26% and 14%, respectively. Residual plasma viremia levels positively correlated with cell-associated HIV RNA and DNA. The performance characteristics of the replicate Aptima assay support its use for quantifying residual plasma viremia to study the latent HIV reservoir and cure interventions.
Keywords: human immunodeficiency virus; latent reservoir; quantification; residual RNA; ultrasensitive viral load; viremia.
Copyright © 2020 Bakkour et al.
Figures

Similar articles
-
Automated Multireplicate Quantification of Persistent HIV-1 Viremia in Individuals on Antiretroviral Therapy.J Clin Microbiol. 2020 Nov 18;58(12):e01442-20. doi: 10.1128/JCM.01442-20. Print 2020 Nov 18. J Clin Microbiol. 2020. PMID: 32967899 Free PMC article.
-
Quantification of HIV-DNA and residual viremia in patients starting ART by droplet digital PCR: Their dynamic decay and correlations with immunological parameters and virological success.J Clin Virol. 2019 Aug;117:61-67. doi: 10.1016/j.jcv.2019.06.004. Epub 2019 Jun 14. J Clin Virol. 2019. PMID: 31229934
-
Size, Composition, and Evolution of HIV DNA Populations during Early Antiretroviral Therapy and Intensification with Maraviroc.J Virol. 2018 Jan 17;92(3):e01589-17. doi: 10.1128/JVI.01589-17. Print 2018 Feb 1. J Virol. 2018. PMID: 29142136 Free PMC article. Clinical Trial.
-
Potential implication of residual viremia in patients on effective antiretroviral therapy.AIDS Res Hum Retroviruses. 2015 Jan;31(1):25-35. doi: 10.1089/AID.2014.0194. AIDS Res Hum Retroviruses. 2015. PMID: 25428885 Free PMC article. Review.
-
Molecular biological assessment methods and understanding the course of the HIV infection.APMIS Suppl. 2003;(114):1-37. APMIS Suppl. 2003. PMID: 14626050 Review.
Cited by
-
Automated Multireplicate Quantification of Persistent HIV-1 Viremia in Individuals on Antiretroviral Therapy.J Clin Microbiol. 2020 Nov 18;58(12):e01442-20. doi: 10.1128/JCM.01442-20. Print 2020 Nov 18. J Clin Microbiol. 2020. PMID: 32967899 Free PMC article.
-
The contemporary immunoassays for HIV diagnosis: a concise overview.Asian Biomed (Res Rev News). 2023 Aug 7;17(1):3-12. doi: 10.2478/abm-2023-0038. eCollection 2023 Feb. Asian Biomed (Res Rev News). 2023. PMID: 37551202 Free PMC article. Review.
-
Validation of digital droplet PCR assays for cell-associated HIV-1 DNA, HIV-1 2-LTR circle, and HIV-1 unspliced RNA for clinical studies in HIV-1 cure research.J Clin Virol. 2024 Feb;170:105632. doi: 10.1016/j.jcv.2023.105632. Epub 2023 Dec 7. J Clin Virol. 2024. PMID: 38113685 Free PMC article.
-
Use of laboratory-developed assays in global HIV-1 treatment-monitoring and research.Sci Rep. 2023 Mar 20;13(1):4578. doi: 10.1038/s41598-023-31103-y. Sci Rep. 2023. PMID: 36941272 Free PMC article. Review.
-
Blood Center Testing Allows the Detection and Rapid Treatment of Acute and Recent HIV Infection.Viruses. 2022 Oct 23;14(11):2326. doi: 10.3390/v14112326. Viruses. 2022. PMID: 36366424 Free PMC article.
References
-
- Gandhi RT, Bosch RJ, Aga E, Bedison MA, Bastow B, Schmitz JL, Siliciano JD, Siliciano RF, Eron JJ, Mellors JW, the ACTG A5173 team. 2013. Residual plasma viraemia and infectious HIV-1 recovery from resting memory CD4 cells in patients on antiretroviral therapy: results from ACTG A5173. Antivir Ther 18:607–613. doi:10.3851/IMP2543. - DOI - PMC - PubMed
-
- Hong F, Jacobs JL, Aga E, Cillo AR, Fyne E, Koontz DL, Zheng L, Mellors JW. 2018. Associations between HIV-1 DNA copy number, proviral transcriptional activity, and plasma viremia in individuals off or on suppressive antiretroviral therapy. Virology 521:51–57. doi:10.1016/j.virol.2018.05.018. - DOI - PMC - PubMed
-
- Eriksson S, Graf EH, Dahl V, Strain MC, Yukl SA, Lysenko ES, Bosch RJ, Lai J, Chioma S, Emad F, Abdel-Mohsen M, Hoh R, Hecht F, Hunt P, Somsouk M, Wong J, Johnston R, Siliciano RF, Richman DD, O'Doherty U, Palmer S, Deeks SG, Siliciano JD. 2013. Comparative analysis of measures of viral reservoirs in HIV-1 eradication studies. PLoS Pathog 9:e1003174. doi:10.1371/journal.ppat.1003174. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

