Stories to Communicate Individual Risk for Opioid Prescriptions for Back and Kidney Stone Pain: Protocol for the Life STORRIED Multicenter Randomized Clinical Trial
- PMID: 32969832
- PMCID: PMC7545334
- DOI: 10.2196/19496
Stories to Communicate Individual Risk for Opioid Prescriptions for Back and Kidney Stone Pain: Protocol for the Life STORRIED Multicenter Randomized Clinical Trial
Abstract
Background: Prescription opioid misuse in the United States is a devastating public health crisis; many chronic opioid users were originally prescribed this class of medication for acute pain. Video narrative-enhanced risk communication may improve patient outcomes, such as knowledge of opioid risk and opioid use behaviors after an episode of acute pain.
Objective: Our objective is to assess the effect of probabilistic and narrative-enhanced opioid risk communication on patient-reported outcomes, including knowledge, opioid use, and patient preferences, for patients who present to emergency departments with back pain and kidney stone pain.
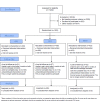
Methods: This is a multisite randomized controlled trial. Patients presenting to the acute care facilities of four geographically and ethnically diverse US hospital centers with acute renal colic pain or musculoskeletal back and/or neck pain are eligible for this randomized controlled trial. A control group of patients receiving general risk information is compared to two intervention groups: one receiving the risk information sheet plus an individualized, visual probabilistic Opioid Risk Tool (ORT) and another receiving the risk information sheet plus a video narrative-enhanced probabilistic ORT. We will study the effect of probabilistic and narrative-enhanced opioid risk communication on the following: risk awareness and recall at 14 days postenrollment, reduced use or preferences for opioids after the emergency department episode, and alignment with patient preference and provider prescription. To assess these outcomes, we administer baseline patient surveys during acute care admission and follow-up surveys at predetermined times during the 3 months after discharge.
Results: A total of 1302 patients were enrolled over 24 months. The mean age of the participants was 40 years (SD 14), 692 out of 1302 (53.15%) were female, 556 out of 1302 (42.70%) were White, 498 out of 1302 (38.25%) were Black, 1002 out of 1302 (76.96%) had back pain, and 334 out of 1302 (25.65%) were at medium or high risk. Demographics and ORT scores were equally distributed across arms.
Conclusions: This study seeks to assess the potential clinical role of narrative-enhanced, risk-informed communication for acute pain management in acute care settings. This paper outlines the protocol used to implement the study and highlights crucial methodological, statistical, and stakeholder involvement as well as dissemination considerations.
Trial registration: ClinicalTrials.gov NCT03134092; https://clinicaltrials.gov/ct2/show/NCT03134092.
International registered report identifier (irrid): DERR1-10.2196/19496.
Keywords: acute pain: opioid risk; musculoskeletal back pain; narratives; opioid misuse; prescription opioids; probabilistic risk tool; randomized controlled trial; renal colic.
©Zachary F Meisel, Erica B Goldberg, Abby R Dolan, Esha Bansal, Karin V Rhodes, Erik P Hess, Carolyn C Cannuscio, Marilyn M Schapira, Jeanmarie Perrone, Melissa A Rodgers, Michael M Zyla, Jeffrey J Bell, Sharon McCollum, Frances S Shofer. Originally published in JMIR Research Protocols (http://www.researchprotocols.org), 24.09.2020.
Conflict of interest statement
Conflicts of Interest: None declared.
Figures





References
-
- Overdose death rates. National Institutes of Health, National Institute on Drug Abuse. 2020. Mar 10, [2019-09-15]. https://www.drugabuse.gov/related-topics/trends-statistics/overdose-deat....
-
- Florence CS, Zhou C, Luo F, Xu L. The economic burden of prescription opioid overdose, abuse, and dependence in the United States, 2013. Med Care. 2016 Oct;54(10):901–906. doi: 10.1097/MLR.0000000000000625. http://europepmc.org/abstract/MED/27623005 - DOI - PMC - PubMed
-
- Opioid overdose overview. Centers for Disease Control and Prevention. 2020. Mar 19, [2019-09-15]. https://www.cdc.gov/drugoverdose/data/prescribing/overview.html.
Associated data
LinkOut - more resources
Full Text Sources
Medical

