Flavopiridol causes cell cycle inhibition and demonstrates anti-cancer activity in anaplastic thyroid cancer models
- PMID: 32970704
- PMCID: PMC7514001
- DOI: 10.1371/journal.pone.0239315
Flavopiridol causes cell cycle inhibition and demonstrates anti-cancer activity in anaplastic thyroid cancer models
Abstract
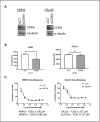
Anaplastic thyroid cancer (ATC) is a rare, but nearly uniformly fatal disease that is typically resistant to chemotherapy and radiation. Alternative strategies to target this cancer at a molecular level are necessary in order to improve dismal outcomes for ATC patients. We examined the effects of flavopiridol, a CDK inhibitor, in a panel of ATC cell lines. When cell lines were treated over a ten-point concentration range, CAL62, KMH2 and BHT-101 cell lines had a sub micromolar half-maximal inhibitory concentration, while no effect was seen in the non-cancerous cell line IMR-90. Flavopiridol treatment resulted in decreased levels of the cell cycle proteins CDK9 and MCL1, and induced cell cycle arrest. Flavopiridol also decreased the in vitro ability of ATC cells to form colonies and impeded migration using a transwell migration assay. In vivo, flavopiridol decreased tumor weight and tumor volume over time in a patient-derived xenograft model of ATC. Given the observed in vitro and in vivo activity, flavopiridol warrants further investigation for treatment of ATC.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



References
-
- Giuffrida D, Gharib H. Anaplastic thyroid carcinoma: Current diagnosis and treatment. 2000; 1–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

