Hydroxychloroquine levels in patients with systemic lupus erythematosus: whole blood is preferable but serum levels also detect non-adherence
- PMID: 32977856
- PMCID: PMC7517694
- DOI: 10.1186/s13075-020-02291-z
Hydroxychloroquine levels in patients with systemic lupus erythematosus: whole blood is preferable but serum levels also detect non-adherence
Abstract
Background: Hydroxychloroquine (HCQ) levels can be measured in both serum and whole blood. No cut-off point for non-adherence has been established in serum nor have these methods ever been compared. The aims of this study were to compare these two approaches and determine if serum HCQ cut-off points can be established to identify non-adherent patients.
Methods: HCQ levels were measured in serum and whole blood from 573 patients with systemic lupus erythematosus (SLE). The risk factors for active SLE (SLEDAI score > 4) were identified by multiple logistic regression. Serum HCQ levels were measured in 68 additional patients known to be non-adherent, i.e. with whole-blood HCQ < 200 ng/mL.
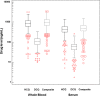
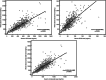
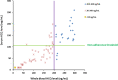
Results: The mean (± SD) HCQ levels were 469 ± 223 ng/mL in serum and 916 ± 449 ng/mL in whole blood. The mean ratio of serum/whole-blood HCQ levels was 0.53 ± 0.15. In the multivariate analysis, low whole-blood HCQ levels (P = 0.023), but not serum HCQ levels, were independently associated with active SLE. From the mean serum/whole-blood level ratio, a serum HCQ level of 106 ng/mL was extrapolated as the corresponding cut-off to identify non-adherent patients with a sensitivity of 0.87 (95% CI 0.76-0.94) and specificity of 0.89 (95% CI 0.72-0.98). All serum HCQ levels of patients with whole-blood HCQ below the detectable level (< 20 ng/mL) were also undetectable (< 20 ng/mL).
Conclusions: These data suggest that whole blood is better than serum for assessing the pharmacokinetic/pharmacodynamic relation of HCQ. Our results support the use of serum HCQ levels to assess non-adherence when whole blood is unavailable.
Keywords: Adherence; Drug monitoring; Hydroxychloroquine; Serum; Systemic lupus erythematosus.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

References
-
- Geraldino-Pardilla L, Perel-Winkler A, Miceli J, Neville K, Danias G, Nguyen S, et al. Association between hydroxychloroquine levels and disease activity in a predominantly Hispanic systemic lupus erythematosus cohort. Lupus. 2019;28(7):862–7. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

