Nociceptin/orphanin FQ neurons in the Arcuate Nucleus and Ventral Tegmental Area Act via Nociceptin Opioid Peptide Receptor Signaling to Inhibit Proopiomelanocortin and A10 Dopamine Neurons and Thereby Modulate Ingestion of Palatable Food
- PMID: 32979341
- PMCID: PMC7736116
- DOI: 10.1016/j.physbeh.2020.113183
Nociceptin/orphanin FQ neurons in the Arcuate Nucleus and Ventral Tegmental Area Act via Nociceptin Opioid Peptide Receptor Signaling to Inhibit Proopiomelanocortin and A10 Dopamine Neurons and Thereby Modulate Ingestion of Palatable Food
Abstract
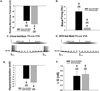
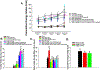
The neuropeptide nociceptin/orphanin FQ (N/OFQ) inhibits neuronal activity via its cognate nociceptin opioid peptide (NOP) receptor throughout the peripheral and central nervous systems, including those areas involved in the homeostatic and hedonic regulation of energy homeostasis. We thus tested the hypothesis that N/OFQ neurons in the hypothalamic arcuate nucleus (ARC) and ventral tegmental area (VTA) act via NOP receptor signaling to inhibit nearby anorexigenic proopiomelanocortin (POMC) and A10 dopamine neuronal excitability, respectively, and thereby modulate ingestion of palatable food. Electrophysiologic recordings were performed in slices prepared from transgenic male and ovariectomized (OVX) female N/OFQ-cre/enhanced green fluorescent protein-POMC, N/OFQ-cre and tyrosine hydroxylase-cre animals to see if optogenetically-stimulated peptide release from N/OFQ neurons could directly inhibit these neuronal populations. Binge-feeding behavioral experiments were also conducted where animals were exposed to a high-fat-diet (HFD) for one hour each day for five days and monitored for energy intake. Photostimulation of ARC and VTA N/OFQ neurons produces an outward current in POMC and A10 dopamine neurons receiving input from these cells. This is associated with a hyperpolarization and decreased firing. These features are also sex hormone- and diet-dependent; with estradiol-treated slices from OVX females being less sensitive, and obese males being more sensitive, to N/OFQ. Limited access to HFD causes a dramatic escalation in consumption, such that animals eat 25-45% of their daily intake during that one-hour exposure. Moreover, the NOP receptor-mediated regulation of these energy balance circuits are engaged, as N/OFQ injected directly into the VTA or ARC respectively diminishes or potentiates this binge-like increase in a manner heightened by diet-induced obesity or dampened by estradiol in females. Collectively, these findings provide key support for the idea that N/OFQ regulates appetitive behavior in sex-, site- and diet-specific ways, along with important insights into aberrant patterns of feeding behavior pertinent to the pathogenesis of food addiction.
Keywords: A(10) dopamine neurons; ARC; Nociceptin; POMC; VTA; energy balance; food addiction; sex differences.
Copyright © 2020 Elsevier Inc. All rights reserved.
Conflict of interest statement
Disclosure Statement
The authors have no conflicts of interest to declare.
Figures

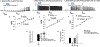



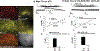
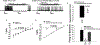
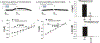
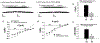
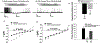

Similar articles
-
Nociceptin/orphanin FQ modulates energy homeostasis through inhibition of neurotransmission at VMN SF-1/ARC POMC synapses in a sex- and diet-dependent manner.Biol Sex Differ. 2019 Feb 12;10(1):9. doi: 10.1186/s13293-019-0220-3. Biol Sex Differ. 2019. PMID: 30755252 Free PMC article.
-
The vital role of arcuate nociceptin/orphanin FQ neurones in mounting an oestradiol-dependent adaptive response to negative energy balance via inhibition of nearby proopiomelanocortin neurones.J Physiol. 2022 Nov;600(22):4939-4961. doi: 10.1113/JP283378. Epub 2022 Oct 28. J Physiol. 2022. PMID: 36217719 Free PMC article.
-
The biology of Nociceptin/Orphanin FQ (N/OFQ) related to obesity, stress, anxiety, mood, and drug dependence.Pharmacol Ther. 2014 Mar;141(3):283-99. doi: 10.1016/j.pharmthera.2013.10.011. Epub 2013 Nov 1. Pharmacol Ther. 2014. PMID: 24189487 Free PMC article. Review.
-
Differential Modulation of Ventral Tegmental Area Circuits by the Nociceptin/Orphanin FQ System.eNeuro. 2020 Oct 19;7(5):ENEURO.0376-19.2020. doi: 10.1523/ENEURO.0376-19.2020. Print 2020 Jul/Aug. eNeuro. 2020. PMID: 32747458 Free PMC article.
-
Nociceptin/orphanin FQ receptor antagonists as innovative antidepressant drugs.Pharmacol Ther. 2013 Oct;140(1):10-25. doi: 10.1016/j.pharmthera.2013.05.008. Epub 2013 May 24. Pharmacol Ther. 2013. PMID: 23711793 Review.
Cited by
-
Neurotransmitters crosstalk and regulation in the reward circuit of subjects with behavioral addiction.Front Psychiatry. 2025 Jan 14;15:1439727. doi: 10.3389/fpsyt.2024.1439727. eCollection 2024. Front Psychiatry. 2025. PMID: 39876994 Free PMC article. Review.
-
Adaptive Changes in the Central Control of Energy Homeostasis Occur in Response to Variations in Energy Status.Int J Mol Sci. 2021 Mar 8;22(5):2728. doi: 10.3390/ijms22052728. Int J Mol Sci. 2021. PMID: 33800452 Free PMC article. Review.
-
Optogenetics in oral and craniofacial research.J Zhejiang Univ Sci B. 2024 Aug 15;25(8):656-671. doi: 10.1631/jzus.B2300322. J Zhejiang Univ Sci B. 2024. PMID: 39155779 Free PMC article. Review.
-
Septo-hypothalamic regulation of binge-like alcohol consumption by the nociceptin system.Cell Rep. 2025 Apr 22;44(4):115482. doi: 10.1016/j.celrep.2025.115482. Epub 2025 Mar 27. Cell Rep. 2025. PMID: 40153436 Free PMC article.
-
Pharmacological blockage of NOP receptors decreases ventral tegmental area dopamine neuronal activity through GABAB receptor-mediated mechanism.Neuropharmacology. 2024 May 1;248:109866. doi: 10.1016/j.neuropharm.2024.109866. Epub 2024 Feb 15. Neuropharmacology. 2024. PMID: 38364970 Free PMC article.
References
-
- Di Marzo V, Ligresti A, Cristino L The endocannabinoid system as a link between homeostatic and hedonic pathways involved in energy balance regulation. Int. J. Obesity 2009,33:S18–S24. - PubMed
-
- Volkow ND, Wise RA, Baler R The dopamine motive system: implications for drug and food addiction. Nat Rev Neurosci. 2017,18:741–52. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

