Knockdown of lncRNA HOXA-AS3 Suppresses the Progression of Atherosclerosis via Sponging miR-455-5p
- PMID: 32982172
- PMCID: PMC7490108
- DOI: 10.2147/DDDT.S249830
Knockdown of lncRNA HOXA-AS3 Suppresses the Progression of Atherosclerosis via Sponging miR-455-5p
Abstract
Background: Atherosclerosis can lead to multiple cardiovascular diseases, especially myocardial infarction. Long noncoding RNAs (lncRNAs) are involved in multiple diseases, including atherosclerosis. LncRNA HOXA-AS3 was found to be notably upregulated in atherosclerosis. However, the biological function of HOXA-AS3 during the occurrence and development of atherosclerosis remains unclear.
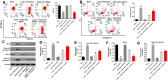
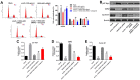
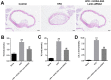
Materials and methods: Human vascular endothelial cells (HUVECs) were treated with oxidized low-density lipoprotein (oxLDL) to mimic atherosclerosis in vitro. Gene and protein expressions in HUVECs were detected by RT-qPCR and Western blot, respectively. Cell proliferation was tested by CCK-8 and Ki67 staining. Cell apoptosis and cycle were measured by flow cytometry. Additionally, the correlation between HOXA-AS3 and miR-455-5p was confirmed by dual luciferase report assay and RNA pull-down. Finally, in vivo model of atherosclerosis was established to confirm the function of HOXA-AS3 during the development of atherosclerosis in vivo.
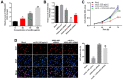
Results: LncRNA HOXA-AS3 was upregulated in oxLDL-treated HUVECs. In addition, oxLDL-induced growth inhibition of HUVECs was significantly reversed by knockdown of HOXA-AS3. Consistently, oxLDL notably induced G1 arrest in HUVECs, while this phenomenon was greatly reversed by HOXA-AS3 siRNA. Furthermore, downregulation of HOXA-AS3 notably inhibited the progression of atherosclerosis through mediation of miR-455-5p/p27 Kip1 axis. Besides, silencing of HOXA-AS3 notably relieved the symptom of atherosclerosis in vivo.
Conclusion: Downregulation of HOXA-AS3 significantly suppressed the progression of atherosclerosis via regulating miR-455-5p/p27 Kip1 axis. Thus, HOXA-AS3 might serve as a potential target for the treatment of atherosclerosis.
Keywords: atherosclerosis; lncRNA HOXA-AS3; miR-455-5p; p27 Kip1.
© 2020 Chi et al.
Conflict of interest statement
The authors declared no competing interests in this research.
Figures



Similar articles
-
LncRNA HOXA-AS3 Promotes the Progression of Pulmonary Arterial Hypertension through Mediation of miR-675-3p/PDE5A Axis.Biochem Genet. 2021 Oct;59(5):1158-1172. doi: 10.1007/s10528-021-10053-y. Epub 2021 Mar 9. Biochem Genet. 2021. PMID: 33687636
-
The integrated comprehension of lncRNA HOXA-AS3 implication on human diseases.Clin Transl Oncol. 2022 Dec;24(12):2342-2350. doi: 10.1007/s12094-022-02920-w. Epub 2022 Aug 20. Clin Transl Oncol. 2022. PMID: 35986859 Free PMC article. Review.
-
Long non-coding RNA HOXA-AS3 promotes cell proliferation of oral squamous cell carcinoma through sponging microRNA miR-218-5p.Bioengineered. 2021 Dec;12(1):8724-8737. doi: 10.1080/21655979.2021.1978196. Bioengineered. 2021. PMID: 34698001 Free PMC article.
-
LncRNA HOXA-AS3 promotes the malignancy of glioblastoma through regulating miR-455-5p/USP3 axis.J Cell Mol Med. 2020 Oct;24(20):11755-11767. doi: 10.1111/jcmm.15788. Epub 2020 Sep 11. J Cell Mol Med. 2020. PMID: 32918360 Free PMC article.
-
Tumour-regulatory role of long non-coding RNA HOXA-AS3.Prog Biophys Mol Biol. 2024 Jul;189:13-25. doi: 10.1016/j.pbiomolbio.2024.04.003. Epub 2024 Apr 7. Prog Biophys Mol Biol. 2024. PMID: 38593905 Review.
Cited by
-
LncRNA HOXA-AS3 Promotes the Progression of Pulmonary Arterial Hypertension through Mediation of miR-675-3p/PDE5A Axis.Biochem Genet. 2021 Oct;59(5):1158-1172. doi: 10.1007/s10528-021-10053-y. Epub 2021 Mar 9. Biochem Genet. 2021. PMID: 33687636
-
The integrated comprehension of lncRNA HOXA-AS3 implication on human diseases.Clin Transl Oncol. 2022 Dec;24(12):2342-2350. doi: 10.1007/s12094-022-02920-w. Epub 2022 Aug 20. Clin Transl Oncol. 2022. PMID: 35986859 Free PMC article. Review.
-
LncRNA HOXA-AS3 promotes cell proliferation and invasion via targeting miR-218-5p/FOXP1 axis in osteosarcoma.Sci Rep. 2024 Jul 17;14(1):16581. doi: 10.1038/s41598-024-67596-4. Sci Rep. 2024. PMID: 39019995 Free PMC article.
-
Upgulation of lncRNA GASL1 inhibits atherosclerosis by regulating miR-106a/LKB1 axis.BMC Cardiovasc Disord. 2023 Jan 10;23(1):11. doi: 10.1186/s12872-023-03038-9. BMC Cardiovasc Disord. 2023. PMID: 36627571 Free PMC article.
-
Clinical Values and Underlying Mechanism Analysis of Serum miR-455-5p in Carotid Artery Stenosis.J Inflamm Res. 2022 May 30;15:3207-3217. doi: 10.2147/JIR.S362774. eCollection 2022. J Inflamm Res. 2022. PMID: 35668916 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

