From Traditional to Targeted Immunotherapy in Myasthenia Gravis: Prospects for Research
- PMID: 32982957
- PMCID: PMC7492201
- DOI: 10.3389/fneur.2020.00981
From Traditional to Targeted Immunotherapy in Myasthenia Gravis: Prospects for Research
Abstract
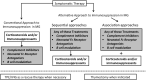
Treatment of Myasthenia Gravis (MG) is still based on non-specific immunosuppression. Long-term high dose corticosteroids is still a major cause of side effects, in young as well as in elderly patients in whom comorbidities further increase the burden of chronic immunosuppression. Moreover, awareness of the limits of traditional therapies has led to the concept of "refractory MG." The therapeutic approach to MG is therefore progressively evolving from the classic combination of corticosteroids and immunosuppressive drugs to new biological compounds targeting different immunopathological steps. Killing of B cells with Rituximab has been proposed and tested with positive results, particularly in patients with MuSK-associated MG. Therapeutic monoclonals against B cells at different stages of their maturation, or against molecules involved in B cell activation and function, represent a new area for further investigation. A differently targeted approach involved Eculizumab, a monoclonal antibody preventing the formation of C59b-induced MAC causing destruction of the neuromuscular junction. Data from clinical trials led to the approval of Eculizumab in the United States and Europe for MG. Since Eculizumab is a complement-targeted therapy, its use is limited to anti-acetylcholine receptor-associated MG, since anti-MuSK antibodies belong to IgG4 subclass and do not fix complement. Several anti-complement compounds are under investigation. An even more recent approach is the interference with the neonatal Fc receptor leading to a rapid reduction of circulating IgGs and hence of specific autoantibodies, an approach suitable for both anti-acetylcholine- and MuSK-associated MG. The investigation of compounds that selectively target the immune system will stimulate the search for specific biomarkers of disease activity and response to treatment, setting the basis for personalized medicine in MG.
Keywords: Eculizumab; Fc receptor; Rituximab; autoimmunity; clinical trials; complement; monoclonal antibodies; myasthenia gravis.
Copyright © 2020 Mantegazza and Antozzi.
Figures

References
Publication types
LinkOut - more resources
Full Text Sources
Medical

