Respiratory CO2 Combined With a Blend of Volatiles Emitted by Endophytic Serendipita Strains Strongly Stimulate Growth of Arabidopsis Implicating Auxin and Cytokinin Signaling
- PMID: 32983211
- PMCID: PMC7492573
- DOI: 10.3389/fpls.2020.544435
Respiratory CO2 Combined With a Blend of Volatiles Emitted by Endophytic Serendipita Strains Strongly Stimulate Growth of Arabidopsis Implicating Auxin and Cytokinin Signaling
Abstract
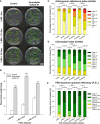
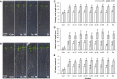
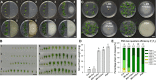
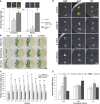
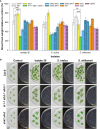
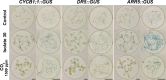
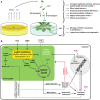
Rhizospheric microorganisms can alter plant physiology and morphology in many different ways including through the emission of volatile organic compounds (VOCs). Here we demonstrate that VOCs from beneficial root endophytic Serendipita spp. are able to improve the performance of in vitro grown Arabidopsis seedlings, with an up to 9.3-fold increase in plant biomass. Additional changes in VOC-exposed plants comprised petiole elongation, epidermal cell and leaf area expansion, extension of the lateral root system, enhanced maximum quantum efficiency of photosystem II (Fv/Fm), and accumulation of high levels of anthocyanin. Notwithstanding that the magnitude of the effects was highly dependent on the test system and cultivation medium, the volatile blends of each of the examined strains, including the references S. indica and S. williamsii, exhibited comparable plant growth-promoting activities. By combining different approaches, we provide strong evidence that not only fungal respiratory CO2 accumulating in the headspace, but also other volatile compounds contribute to the observed plant responses. Volatile profiling identified methyl benzoate as the most abundant fungal VOC, released especially by Serendipita cultures that elicit plant growth promotion. However, under our experimental conditions, application of methyl benzoate as a sole volatile did not affect plant performance, suggesting that other compounds are involved or that the mixture of VOCs, rather than single molecules, accounts for the strong plant responses. Using Arabidopsis mutant and reporter lines in some of the major plant hormone signal transduction pathways further revealed the involvement of auxin and cytokinin signaling in Serendipita VOC-induced plant growth modulation. Although we are still far from translating the current knowledge into the implementation of Serendipita VOCs as biofertilizers and phytostimulants, volatile production is a novel mechanism by which sebacinoid fungi can trigger and control biological processes in plants, which might offer opportunities to address agricultural and environmental problems in the future.
Keywords: Piriformospora; endophytic Sebacinales; fungal volatiles; phytohormone signaling; plant growth and development; plant-microbe interactions.
Copyright © 2020 Venneman, Vandermeersch, Walgraeve, Audenaert, Ameye, Verwaeren, Steppe, Van Langenhove, Haesaert and Vereecke.
Figures




Similar articles
-
Volatile organic compounds emitted by Trichoderma species mediate plant growth.Fungal Biol Biotechnol. 2016 Sep 29;3:7. doi: 10.1186/s40694-016-0025-7. eCollection 2016. Fungal Biol Biotechnol. 2016. PMID: 28955466 Free PMC article.
-
Plant Growth Promotion by Two Volatile Organic Compounds Emitted From the Fungus Cladosporium halotolerans NGPF1.Front Plant Sci. 2021 Dec 3;12:794349. doi: 10.3389/fpls.2021.794349. eCollection 2021. Front Plant Sci. 2021. PMID: 34925431 Free PMC article.
-
Red:far-red light conditions affect the emission of volatile organic compounds from barley (Hordeum vulgare), leading to altered biomass allocation in neighbouring plants.Ann Bot. 2015 May;115(6):961-70. doi: 10.1093/aob/mcv036. Epub 2015 Apr 7. Ann Bot. 2015. PMID: 25851141 Free PMC article.
-
Microbial volatiles as plant growth inducers.Microbiol Res. 2018 Mar;208:63-75. doi: 10.1016/j.micres.2018.01.002. Epub 2018 Jan 31. Microbiol Res. 2018. PMID: 29551213 Review.
-
Role of Phytohormones in Piriformospora indica-Induced Growth Promotion and Stress Tolerance in Plants: More Questions Than Answers.Front Microbiol. 2018 Jul 31;9:1646. doi: 10.3389/fmicb.2018.01646. eCollection 2018. Front Microbiol. 2018. PMID: 30140257 Free PMC article. Review.
Cited by
-
Streptomyces-triggered coordination between rhizosphere microbiomes and plant transcriptome enables watermelon Fusarium wilt resistance.Microb Biotechnol. 2024 Mar;17(3):e14435. doi: 10.1111/1751-7915.14435. Microb Biotechnol. 2024. PMID: 38465781 Free PMC article.
-
Friends in Arms: Flavonoids and the Auxin/Cytokinin Balance in Terrestrialization.Plants (Basel). 2023 Jan 23;12(3):517. doi: 10.3390/plants12030517. Plants (Basel). 2023. PMID: 36771601 Free PMC article. Review.
-
How Do Plant Growth-Promoting Bacteria Use Plant Hormones to Regulate Stress Reactions?Plants (Basel). 2024 Aug 26;13(17):2371. doi: 10.3390/plants13172371. Plants (Basel). 2024. PMID: 39273855 Free PMC article. Review.
-
Optimal culture conditions of Piriformospora indica for volatile compound release to promote effective plant growth.Plant Biotechnol (Tokyo). 2023 Mar 25;40(1):117-121. doi: 10.5511/plantbiotechnology.22.1221a. Plant Biotechnol (Tokyo). 2023. PMID: 38213916 Free PMC article.
-
Mechanisms in Growth-Promoting of Cucumber by the Endophytic Fungus Chaetomium globosum Strain ND35.J Fungi (Basel). 2022 Feb 11;8(2):180. doi: 10.3390/jof8020180. J Fungi (Basel). 2022. PMID: 35205933 Free PMC article.
References
-
- Achatz B., von Rüden S., Andrade D., Neumann E., Pons-Kühnemann J., Kogel K. H., et al. (2010). Root colonization by Piriformospora indica enhances grain yield in barley under diverse nutrient regimes by accelerating plant development. Plant Soil 333, 59–70. 10.1007/s11104-010-03190 - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous

