Patient-Reported Outcomes from a 1-Year, Real-World, Head-to-Head Comparison of OnabotulinumtoxinA and Topiramate for Headache Prevention in Adults With Chronic Migraine
- PMID: 32985341
- PMCID: PMC7536482
- DOI: 10.1177/2150132720959936
Patient-Reported Outcomes from a 1-Year, Real-World, Head-to-Head Comparison of OnabotulinumtoxinA and Topiramate for Headache Prevention in Adults With Chronic Migraine
Abstract
Introduction/objective: Chronic migraine (CM) is associated with impaired health-related quality of life and substantial socioeconomic burden, but many people with CM are underdiagnosed and do not receive appropriate preventive treatment. OnabotulinumtoxinA and topiramate have demonstrated efficacy (treatment benefit under ideal conditions) for the prevention of headaches in people with CM in clinical trials, but real-world studies suggest markedly different clinical effectiveness (treatment benefit based on a blend of efficacy and tolerability). This study sought to evaluate patient-reported outcomes (PROs) of onabotulinumtoxinA versus topiramate immediate release for people with CM.
Methods: FORWARD was a prospective, multicenter, randomized, parallel-group, open-label, phase 4 study comparing onabotulinumtoxinA 155 U every 12 weeks with topiramate 50 to 100 mg/day for ≤36 weeks in people with CM. PROs measured included the Headache Impact Test (HIT-6), 9-item Patient Health Questionnaire Quick Depression Assessment (PHQ-9), Work Productivity and Activity Impairment Questionnaire: Specific Health Problem (WPAI:SHP), and Functional Impact of Migraine Questionnaire (FIMQ).
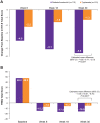
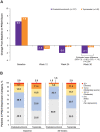
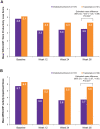
Results: A total of 282 patients were randomized and treated with onabotulinumtoxinA (n = 140) or topiramate (n = 142). From baseline to week 30, mean HIT-6 test scores improved significantly in patients taking onabotulinumtoxinA compared with topiramate (P < .001). Improvements in depression over time were observed via larger changes in PHQ-9 scores with onabotulinumtoxinA than topiramate (P < .001). Work productivity assessed via WPAI:SHP scores revealed significant improvements with onabotulinumtoxinA versus topiramate in Work Productivity Loss (P = .024) and Activity Impairment (P < .001) domains. Results from the FIMQ also revealed a larger reduction from baseline with onabotulinumtoxinA vs topiramate (P < .0001).
Conclusion: OnabotulinumtoxinA treatment had more favorable real-world effectiveness than topiramate on depression, headache impact, functioning and daily living, activity, and work productivity. The overall study results suggest that the beneficial effects on a range of PROs are the result of improved effectiveness when onabotulinumtoxinA is used as preventive treatment for CM.
Trial registration: clinicaltrials.gov: NCT02191579; https://clinicaltrials.gov/ct2/show/NCT02191579.
Keywords: activities of daily living; anxiety; depression; headache; migraine disorders; onabotulinumtoxinA; patient health questionnaire; patient-reported outcome measures; quality of life; topiramate; treatment outcome.
Conflict of interest statement
Figures
Similar articles
-
FORWARD Study: Evaluating the Comparative Effectiveness of OnabotulinumtoxinA and Topiramate for Headache Prevention in Adults With Chronic Migraine.Headache. 2019 Nov;59(10):1700-1713. doi: 10.1111/head.13653. Epub 2019 Sep 26. Headache. 2019. PMID: 31559634 Free PMC article. Clinical Trial.
-
A double-blind comparison of onabotulinumtoxina (BOTOX) and topiramate (TOPAMAX) for the prophylactic treatment of chronic migraine: a pilot study.Headache. 2009 Nov-Dec;49(10):1466-78. doi: 10.1111/j.1526-4610.2009.01566.x. Headache. 2009. PMID: 19912346 Clinical Trial.
-
An open-label prospective study of the real-life use of onabotulinumtoxinA for the treatment of chronic migraine: the REPOSE study.J Headache Pain. 2019 Mar 7;20(1):26. doi: 10.1186/s10194-019-0976-1. J Headache Pain. 2019. PMID: 30845917 Free PMC article.
-
Effectiveness of onabotulinumtoxinA (BOTOX®) for the preventive treatment of chronic migraine: A meta-analysis on 10 years of real-world data.Cephalalgia. 2022 Dec;42(14):1543-1564. doi: 10.1177/03331024221123058. Epub 2022 Sep 8. Cephalalgia. 2022. PMID: 36081276 Free PMC article.
-
OnabotulinumtoxinA: Discussion of the evidence for effectiveness of OnabotulinumA and its place in chronic migraine treatment.Handb Clin Neurol. 2024;199:87-106. doi: 10.1016/B978-0-12-823357-3.00007-0. Handb Clin Neurol. 2024. PMID: 38307674 Review.
Cited by
-
Effect of Atogepant for Preventive Migraine Treatment on Patient-Reported Outcomes in the Randomized, Double-blind, Phase 3 ADVANCE Trial.Neurology. 2023 Feb 21;100(8):e764-e777. doi: 10.1212/WNL.0000000000201568. Epub 2022 Nov 17. Neurology. 2023. PMID: 36396451 Free PMC article. Clinical Trial.
-
Efficacy of onabotulinumtoxinA treatment in episodic migraine.Front Neurol. 2025 Jan 6;15:1459767. doi: 10.3389/fneur.2024.1459767. eCollection 2024. Front Neurol. 2025. PMID: 39835150 Free PMC article.
-
Physicians' Practice of the Non-Cosmetic Uses of Botulinum Toxin: A Cross-Sectional Study in Saudi Arabia.Cureus. 2022 Jan 17;14(1):e21326. doi: 10.7759/cureus.21326. eCollection 2022 Jan. Cureus. 2022. PMID: 35186585 Free PMC article.
-
OnabotulinumtoxinA: Still the Present for Chronic Migraine.Toxins (Basel). 2023 Jan 10;15(1):59. doi: 10.3390/toxins15010059. Toxins (Basel). 2023. PMID: 36668879 Free PMC article. Review.
-
Validation and meaningful within-patient change in work productivity and activity impairment questionnaire (WPAI) for episodic or chronic migraine.J Patient Rep Outcomes. 2023 Apr 4;7(1):34. doi: 10.1186/s41687-023-00552-4. J Patient Rep Outcomes. 2023. PMID: 37016181 Free PMC article. Clinical Trial.
References
-
- Schwedt TJ. Chronic migraine. BMJ. 2014;348:g1416. - PubMed
-
- American Headache Society. The American Headache Society position statement on integrating new migraine treatments into clinical practice. Headache. 2019;59:1-18. - PubMed
-
- Silberstein SD. Practice parameter: evidence-based guidelines for migraine headache (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology. 2000;55:754-762. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

