Intercellular Transmission of Naked Viruses through Extracellular Vesicles: Focus on Polyomaviruses
- PMID: 32993049
- PMCID: PMC7599864
- DOI: 10.3390/v12101086
Intercellular Transmission of Naked Viruses through Extracellular Vesicles: Focus on Polyomaviruses
Abstract
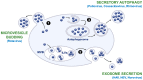
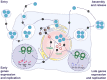
Extracellular vesicles have recently emerged as a novel mode of viral transmission exploited by naked viruses to exit host cells through a nonlytic pathway. Extracellular vesicles can allow multiple viral particles to collectively traffic in and out of cells, thus enhancing the viral fitness and diversifying the transmission routes while evading the immune system. This has been shown for several RNA viruses that belong to the Picornaviridae, Hepeviridae, Reoviridae, and Caliciviridae families; however, recent studies also demonstrated that the BK and JC viruses, two DNA viruses that belong to the Polyomaviridae family, use a similar strategy. In this review, we provide an update on recent advances in understanding the mechanisms used by naked viruses to hijack extracellular vesicles, and we discuss the implications for the biology of polyomaviruses.
Keywords: BKPyV; JCPyV; MCPyV; Polyomavirus; SV40; TSPyV; en bloc transmission; extracellular vesicles; neutralizing antibodies.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Robinson S.M., Tsueng G., Sin J., Mangale V., Rahawi S., McIntyre L.L., Williams W., Kha N., Cruz C., Hancock B.M., et al. Coxsackievirus B exits the host cell in shed microvesicles displaying autophagosomal markers. PLoS Pathog. 2014;10:e1004045. doi: 10.1371/journal.ppat.1004045. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

