Effects of gp120 Inner Domain (ID2) Immunogen Doses on Elicitation of Anti-HIV-1 Functional Fc-Effector Response to C1/C2 (Cluster A) Epitopes in Mice
- PMID: 32998443
- PMCID: PMC7650682
- DOI: 10.3390/microorganisms8101490
Effects of gp120 Inner Domain (ID2) Immunogen Doses on Elicitation of Anti-HIV-1 Functional Fc-Effector Response to C1/C2 (Cluster A) Epitopes in Mice
Abstract
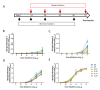
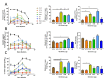
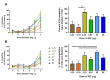
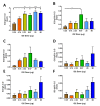
Fc-mediated effector functions of antibodies, including antibody-dependent cytotoxicity (ADCC), have been shown to contribute to vaccine-induced protection from HIV-1 infection, especially those directed against non-neutralizing, CD4 inducible (CD4i) epitopes within the gp120 constant 1 and 2 regions (C1/C2 or Cluster A epitopes). However, recent passive immunization studies have not been able to definitively confirm roles for these antibodies in HIV-1 prevention mostly due to the complications of cross-species Fc-FcR interactions and suboptimal dosing strategies. Here, we use our stabilized gp120 Inner domain (ID2) immunogen that displays the Cluster A epitopes within a minimal structural unit of HIV-1 Env to investigate an immunization protocol that induces a fine-tuned antibody repertoire capable of an effective Fc-effector response. This includes the generation of isotypes and the enhanced antibody specificity known to be vital for maximal Fc-effector activities, while minimizing the induction of isotypes know to be detrimental for these functions. Although our studies were done in in BALB/c mice we conclude that when optimally titrated for the species of interest, ID2 with GLA-SE adjuvant will elicit high titers of antibodies targeting the Cluster A region with potent Fc-mediated effector functions, making it a valuable immunogen candidate for testing an exclusive role of non-neutralizing antibody response in HIV-1 protection in vaccine settings.
Keywords: ADCC; HIV-1; dosing; fc-mediated effector functions; inner domain (ID2) immunogen; isotype; non-neutralizing antibody response.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Incorporating the Cluster A and V1V2 Targets into a Minimal Structural Unit of the HIV-1 Envelope to Elicit a Cross-Clade Response with Potent Fc-Effector Functions.Vaccines (Basel). 2021 Aug 31;9(9):975. doi: 10.3390/vaccines9090975. Vaccines (Basel). 2021. PMID: 34579212 Free PMC article.
-
Induction of Fc-Mediated Effector Functions Against a Stabilized Inner Domain of HIV-1 gp120 Designed to Selectively Harbor the A32 Epitope Region.Front Immunol. 2019 Apr 2;10:677. doi: 10.3389/fimmu.2019.00677. eCollection 2019. Front Immunol. 2019. PMID: 31001276 Free PMC article.
-
Cocrystal Structures of Antibody N60-i3 and Antibody JR4 in Complex with gp120 Define More Cluster A Epitopes Involved in Effective Antibody-Dependent Effector Function against HIV-1.J Virol. 2015 Sep;89(17):8840-54. doi: 10.1128/JVI.01232-15. Epub 2015 Jun 17. J Virol. 2015. PMID: 26085162 Free PMC article.
-
Conformational Masking and Receptor-Dependent Unmasking of Highly Conserved Env Epitopes Recognized by Non-Neutralizing Antibodies That Mediate Potent ADCC against HIV-1.Viruses. 2015 Sep 18;7(9):5115-32. doi: 10.3390/v7092856. Viruses. 2015. PMID: 26393642 Free PMC article. Review.
-
Structural Basis for Epitopes in the gp120 Cluster A Region that Invokes Potent Effector Cell Activity.Viruses. 2019 Jan 16;11(1):69. doi: 10.3390/v11010069. Viruses. 2019. PMID: 30654465 Free PMC article. Review.
Cited by
-
Incorporating the Cluster A and V1V2 Targets into a Minimal Structural Unit of the HIV-1 Envelope to Elicit a Cross-Clade Response with Potent Fc-Effector Functions.Vaccines (Basel). 2021 Aug 31;9(9):975. doi: 10.3390/vaccines9090975. Vaccines (Basel). 2021. PMID: 34579212 Free PMC article.
References
-
- Gresele P., Falcinelli E., Momi S., Francisci D., Baldelli F. Highly active antiretroviral therapy-related mechanisms of endothelial and platelet function alterations. Rev. Cardiovasc. Med. 2014;15(Suppl. 1):S9–S20. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

