Itacitinib (INCB039110), a JAK1 Inhibitor, Reduces Cytokines Associated with Cytokine Release Syndrome Induced by CAR T-cell Therapy
- PMID: 32998963
- PMCID: PMC7895329
- DOI: 10.1158/1078-0432.CCR-20-1739
Itacitinib (INCB039110), a JAK1 Inhibitor, Reduces Cytokines Associated with Cytokine Release Syndrome Induced by CAR T-cell Therapy
Abstract
Purpose: T cells engineered to express a chimeric antigen receptor (CAR) are a promising cancer immunotherapy. Such targeted therapies have shown long-term relapse-free survival in patients with B-cell leukemia and lymphoma. However, cytokine release syndrome (CRS) represents a serious, potentially life-threatening side effect often associated with CAR T-cell therapy. CRS manifests as a rapid (hyper)immune reaction driven by excessive inflammatory cytokine release, including IFNγ and IL6.
Experimental design: Many cytokines implicated in CRS are known to signal through the JAK-STAT pathway. Here we study the effect of blocking JAK pathway signaling on CAR T-cell proliferation, antitumor activity, and cytokine levels in in vitro and in vivo models.
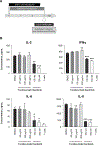
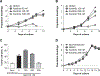
Results: We report that itacitinib, a potent, selective JAK1 inhibitor, was able to significantly and dose-dependently reduce levels of multiple cytokines implicated in CRS in several in vitro and in vivo models. Importantly, we also report that at clinically relevant doses that mimic human JAK1 pharmacologic inhibition, itacitinib did not significantly inhibit proliferation or antitumor killing capacity of three different human CAR T-cell constructs (GD2, EGFR, and CD19). Finally, in an in vivo model, antitumor activity of CD19-CAR T cells adoptively transferred into CD19+ tumor-bearing immunodeficient animals was unabated by oral itacitinib treatment.
Conclusions: Together, these data suggest that itacitinib has potential as a prophylactic agent for the prevention of CAR T cell-induced CRS, and a phase II clinical trial of itacitinib for prevention of CRS induced by CAR T-cell therapy has been initiated (NCT04071366).
©2020 American Association for Cancer Research.
Conflict of interest statement
Figures

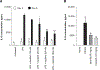

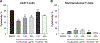

Similar articles
-
Preclinical characterization of itacitinib (INCB039110), a novel selective inhibitor of JAK1, for the treatment of inflammatory diseases.Eur J Pharmacol. 2020 Oct 15;885:173505. doi: 10.1016/j.ejphar.2020.173505. Epub 2020 Aug 28. Eur J Pharmacol. 2020. PMID: 32861662
-
[Establishment of a cytokine release syndrome associated with chimeric antigen receptor T cell treatment in SCID/Beige mice model].Zhonghua Zhong Liu Za Zhi. 2021 Dec 23;43(12):1248-1254. doi: 10.3760/cma.j.cn112152-20190916-00598. Zhonghua Zhong Liu Za Zhi. 2021. PMID: 34915632 Chinese.
-
Individual Patient Data Meta-Analysis from 16 Trials for Safety Factors in Cytokine Release Syndrome After CAR-T Therapy in Patients with Non-Hodgkin Lymphoma (NHL) and Acute Lymphoblastic Leukemia.Adv Ther. 2019 Oct;36(10):2881-2894. doi: 10.1007/s12325-019-01056-8. Epub 2019 Aug 19. Adv Ther. 2019. PMID: 31428935
-
Using JAK inhibitor to treat cytokine release syndrome developed after chimeric antigen receptor T cell therapy for patients with refractory acute lymphoblastic leukemia: A case report.Medicine (Baltimore). 2021 May 14;100(19):e25786. doi: 10.1097/MD.0000000000025786. Medicine (Baltimore). 2021. PMID: 34106613 Free PMC article.
-
Assessment and management of cytokine release syndrome and neurotoxicity following CD19 CAR-T cell therapy.Expert Opin Biol Ther. 2020 Jun;20(6):653-664. doi: 10.1080/14712598.2020.1729735. Epub 2020 Feb 24. Expert Opin Biol Ther. 2020. PMID: 32067497 Free PMC article. Review.
Cited by
-
Hemophagocytic lymphohistiocytosis-like toxicity (carHLH) after CD19-specific CAR T-cell therapy.Br J Haematol. 2021 Aug;194(4):701-707. doi: 10.1111/bjh.17662. Epub 2021 Jul 15. Br J Haematol. 2021. PMID: 34263927 Free PMC article.
-
JAK/STAT of all trades: linking inflammation with cancer development, tumor progression and therapy resistance.Carcinogenesis. 2021 Dec 31;42(12):1411-1419. doi: 10.1093/carcin/bgab075. Carcinogenesis. 2021. PMID: 34415330 Free PMC article.
-
Advances and challenges in CAR-T cell therapy for head and neck squamous cell carcinoma.Biomark Res. 2025 May 1;13(1):69. doi: 10.1186/s40364-025-00783-1. Biomark Res. 2025. PMID: 40312353 Free PMC article. Review.
-
Differential effects of itacitinib, fedratinib, and ruxolitinib in mouse models of hemophagocytic lymphohistiocytosis.Blood. 2024 Jun 6;143(23):2386-2400. doi: 10.1182/blood.2023021046. Blood. 2024. PMID: 38446698 Free PMC article.
-
Identification of genomic determinants contributing to cytokine release in immunotherapies and human diseases.J Transl Med. 2022 Jul 28;20(1):338. doi: 10.1186/s12967-022-03531-3. J Transl Med. 2022. PMID: 35902861 Free PMC article.
References
-
- Schuster SJ, Bishop MR, Tam CS, Waller EK, Borchmann P, McGuirk JP, et al. Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. N Engl J Med 2019;380:45–56. - PubMed
-
- Gill S, Maus MV, Porter DL. Chimeric antigen receptor T cell therapy: 25 years in the making. Blood Rev 2016;30:157–67. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

