Pinealectomy increases thermogenesis and decreases lipogenesis
- PMID: 33000192
- PMCID: PMC7533451
- DOI: 10.3892/mmr.2020.11534
Pinealectomy increases thermogenesis and decreases lipogenesis
Abstract
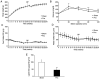
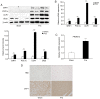
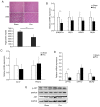
The present study was designed to determine the effects of pineal gland‑derived melatonin on obesity by employing a rat pinealectomy (Pnx) model. After 10 weeks of a high‑fat diet, rats received sham or Pnx surgery followed by a normal chow diet for 10 weeks. Reverse transcription‑quantitative PCR, western blotting analysis, immunohistochemistry and ELISA were used to determine the effects of Pnx. Pnx decreased the expression of melatonin receptor (MTNR)1A and MTNR1B, in brown adipose tissues (BAT) and white adipose tissues (WAT). Pnx rats showed increased insulin sensitivity compared with those that received sham surgery. Leptin levels were significantly decreased in the serum of the Pnx group. In addition, Pnx stimulated thermogenic genes in BAT and attenuated lipogenic genes in both WAT and the liver. Histological analyses revealed a marked decrease in the size of lipid droplets and increased expression of uncoupling protein 1 in BAT. In the liver of the Pnx group, the size and number of lipid droplets had also decreased. In conclusion, the results presented in the current study suggested that Pnx increases thermogenesis in BAT and decreases lipogenesis in WAT and the liver.
Figures


References
-
- Ng M, Fleming T, Robinson M, Thomson B, Graetz N, Margono C, Mullany EC, Biryukov S, Abbafati C, Ferede S, et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: A systematic analysis for the global burden of disease study 2013. Lancet. 2014;384:766–781. doi: 10.1016/S0140-6736(14)60460-8. - DOI - PMC - PubMed
-
- Prospective Studies Collaboration. Whitlock G, Lewington S, Sherliker P, Clarke R, Emberson J, Halsey J, Qizilbash N, Collins R, Peto R. Body-mass index and cause-specific mortality in 900 000 adults: Collaborative analyses of 57 prospective studies. Lancet. 2009;373:1083–1096. doi: 10.1016/S0140-6736(09)60318-4. - DOI - PMC - PubMed
-
- Skrypnik D, Bogdański P, Zawiejska A, Wender-Ożegowska E. Role of gestational weight gain, gestational diabetes, breastfeeding, and hypertension in mother-to-child obesity transmission. Pol Arch Intern Med. 2019;129:267–275. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

