Influenza infection fortifies local lymph nodes to promote lung-resident heterosubtypic immunity
- PMID: 33005934
- PMCID: PMC7534905
- DOI: 10.1084/jem.20200218
Influenza infection fortifies local lymph nodes to promote lung-resident heterosubtypic immunity
Abstract
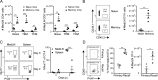
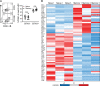
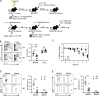
Influenza infection generates tissue-resident memory T cells (TRMs) that are maintained in the lung and can mediate protective immunity to heterologous influenza strains, but the precise mechanisms of local T cell-mediated protection are not well understood. In a murine heterosubtypic influenza challenge model, we demonstrate that protective lung T cell responses derive from both in situ activation of TRMs and the enhanced generation of effector T cells from the local lung draining mediastinal lymph nodes (medLNs). Primary infection fortified the medLNs with an increased number of conventional dendritic cells (cDCs) that mediate enhanced priming of T cells, including those specific for newly encountered epitopes; cDC depletion during the recall response diminished medLN T cell generation and heterosubtypic immunity. Our study shows that during a protective recall response, cDCs in a fortified LN environment enhance the breadth, generation, and tissue migration of effector T cells to augment lung TRM responses.
© 2020 Paik and Farber.
Conflict of interest statement
Disclosures: The authors declare no competing interests exist.
Figures







References
-
- Beura L.K., Mitchell J.S., Thompson E.A., Schenkel J.M., Mohammed J., Wijeyesinghe S., Fonseca R., Burbach B.J., Hickman H.D., Vezys V., et al. . 2018. Intravital mucosal imaging of CD8+ resident memory T cells shows tissue-autonomous recall responses that amplify secondary memory. Nat. Immunol. 19:173–182. 10.1038/s41590-017-0029-3 - DOI - PMC - PubMed
-
- Cunningham N.R., Artim S.C., Fornadel C.M., Sellars M.C., Edmonson S.G., Scott G., Albino F., Mathur A., and Punt J.A.. 2006. Immature CD4+CD8+ thymocytes and mature T cells regulate Nur77 distinctly in response to TCR stimulation. J. Immunol. 177:6660–6666. 10.4049/jimmunol.177.10.6660 - DOI - PubMed
-
- Francis T. 1960. On the doctrine of original antigenic sin. Proc. Am. Philos. Soc. 104:572–578.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

