Targeting the p53-MDM2 pathway for neuroblastoma therapy: Rays of hope
- PMID: 33007410
- PMCID: PMC8351219
- DOI: 10.1016/j.canlet.2020.09.023
Targeting the p53-MDM2 pathway for neuroblastoma therapy: Rays of hope
Abstract
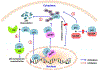
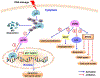
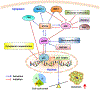
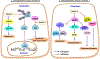
Despite being the subject of extensive research and clinical trials, neuroblastoma remains a major therapeutic challenge in pediatric oncology. The p53 protein is a central safeguard that protects cells against genome instability and malignant transformation. Mutated TP53 (the gene encoding p53) is implicated in many human cancers, but the majority of neuroblastomas have wild type p53 with intact transcriptional function. In fact, the TP53 mutation rate does not exceed 1-2% in neuroblastomas. However, overexpression of the murine double minute 2 (MDM2) gene in neuroblastoma is relatively common, and leads to inhibition of p53. It is also associated with other non-canonical p53-independent functions, including drug resistance and increased translation of MYCN and VEGF mRNA. The p53-MDM2 pathway in neuroblastoma is also modulated at several different molecular levels, including via interactions with other proteins (MYCN, p14ARF). In addition, the overexpression of MDM2 in tumors is linked to a poorer prognosis for cancer patients. Thus, restoring p53 function by inhibiting its interaction with MDM2 is a potential therapeutic strategy for neuroblastoma. A number of p53-MDM2 antagonists have been designed and studied for this purpose. This review summarizes the current understanding of p53 biology and the p53-dependent and -independent oncogenic functions of MDM2 in neuroblastoma, and also the regulation of the p53-MDM2 axis in neuroblastoma. This review also highlights the use of MDM2 as a molecular target for the disease, and describes the MDM2 inhibitors currently being investigated in preclinical and clinical studies. We also briefly explain the various strategies that have been used and future directions to take in the development of effective MDM2 inhibitors for neuroblastoma.
Keywords: Inhibitors; MDM2; Neuroblastoma; Targeted therapy; p53.
Copyright © 2020 Elsevier B.V. All rights reserved.
Conflict of interest statement
Conflict of interest
We wish to confirm that there are no known conflicts of interest associated with this publication and there has been no significant financial support for this work that could have influenced its outcome.
Figures
References
-
- Nakagawara A, Li Y, Izumi H, Muramori K, Inada H, Nishi M, Neuroblastoma, Japanese journal of clinical oncology, 48 (2018) 214–241. - PubMed
-
- Mallepalli S, Gupta MK, Vadde R, Neuroblastoma: An Updated Review on Biology and Treatment, Current drug metabolism, 20 (2019) 1014–1022. - PubMed
-
- Newman EA, Abdessalam S, Aldrink JH, Austin M, Heaton TE, Bruny J, Ehrlich P, Dasgupta R, Baertschiger RM, Lautz TB, Rhee DS, Langham MR Jr., Malek MM, Meyers RL, Nathan JD, Weil BR, Polites S, Madonna MB, Update on neuroblastoma, Journal of pediatric surgery, 54 (2019) 383–389. - PubMed
-
- Gatta G, Botta L, Rossi S, Aareleid T, Bielska-Lasota M, Clavel J, Dimitrova N, Jakab Z, Kaatsch P, Lacour B, Mallone S, Marcos-Gragera R, Minicozzi P, Sánchez-Pérez MJ, Sant M, Santaquilani M, Stiller C, Tavilla A, Trama A, Visser O, Peris-Bonet R, Childhood cancer survival in Europe 1999–2007: results of EUROCARE-5--a population-based study, The Lancet. Oncology, 15 (2014) 35–47. - PubMed
-
- Lacour B, Goujon S, Guissou S, Guyot-Goubin A, Desmée S, Désandes E, Clavel J, Childhood cancer survival in France, 2000–2008, European journal of cancer prevention : the official journal of the European Cancer Prevention Organisation (ECP), 23 (2014) 449–457. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

