Matrix Remodeling and Hyaluronan Production by Myofibroblasts and Cancer-Associated Fibroblasts in 3D Collagen Matrices
- PMID: 33008082
- PMCID: PMC7709683
- DOI: 10.3390/gels6040033
Matrix Remodeling and Hyaluronan Production by Myofibroblasts and Cancer-Associated Fibroblasts in 3D Collagen Matrices
Abstract
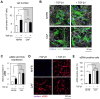
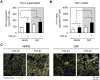
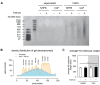
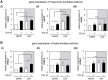
The tumor microenvironment is a key modulator in cancer progression and has become a novel target in cancer therapy. An increase in hyaluronan (HA) accumulation and metabolism can be found in advancing tumor progression and are often associated with aggressive malignancy, drug resistance and poor prognosis. Wound-healing related myofibroblasts or activated cancer-associated fibroblasts (CAF) are assumed to be the major sources of HA. Both cell types are capable to synthesize new matrix components as well as reorganize the extracellular matrix. However, to which extent myofibroblasts and CAF perform these actions are still unclear. In this work, we investigated the matrix remodeling and HA production potential in normal human dermal fibroblasts (NHFB) and CAF in the absence and presence of transforming growth factor beta -1 (TGF-β1), with TGF-β1 being a major factor of regulating fibroblast differentiation. Three-dimensional (3D) collagen matrix was utilized to mimic the extracellular matrix of the tumor microenvironment. We found that CAF appeared to response insensitively towards TGF-β1 in terms of cell proliferation and matrix remodeling when compared to NHFB. In regards of HA production, we found that both cell types were capable to produce matrix bound HA, rather than a soluble counterpart, in response to TGF-β1. However, activated CAF demonstrated higher HA production when compared to myofibroblasts. The average molecular weight of produced HA was found in the range of 480 kDa for both cells. By analyzing gene expression of HA metabolizing enzymes, namely hyaluronan synthase (HAS1-3) and hyaluronidase (HYAL1-3) isoforms, we found expression of specific isoforms in dependence of TGF-β1 present in both cells. In addition, HAS2 and HYAL1 are highly expressed in CAF, which might contribute to a higher production and degradation of HA in CAF matrix. Overall, our results suggested a distinct behavior of NHFB and CAF in 3D collagen matrices in the presence of TGF-β1 in terms of matrix remodeling and HA production pointing to a specific impact on tumor modulation.
Keywords: cancer associated fibroblast; hyaluronan; matrix remodeling; myofibroblast; transforming growth factor beta 1; tumor microenvironment.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Response of fibroblasts to transforming growth factor-β1 on two-dimensional and in three-dimensional hyaluronan hydrogels.Tissue Eng Part A. 2012 Dec;18(23-24):2528-38. doi: 10.1089/ten.TEA.2012.0094. Epub 2012 Aug 21. Tissue Eng Part A. 2012. PMID: 22734649 Free PMC article.
-
Hyaluronan Controls the Deposition of Fibronectin and Collagen and Modulates TGF-β1 Induction of Lung Myofibroblasts.Matrix Biol. 2015 Mar;42:74-92. doi: 10.1016/j.matbio.2014.12.001. Epub 2014 Dec 27. Matrix Biol. 2015. PMID: 25549589 Free PMC article.
-
Regulation of Hyaluronan (HA) Metabolism Mediated by HYBID (Hyaluronan-binding Protein Involved in HA Depolymerization, KIAA1199) and HA Synthases in Growth Factor-stimulated Fibroblasts.J Biol Chem. 2015 Dec 25;290(52):30910-23. doi: 10.1074/jbc.M115.673566. Epub 2015 Oct 30. J Biol Chem. 2015. PMID: 26518873 Free PMC article.
-
Hyaluronan - a functional and structural sweet spot in the tissue microenvironment.Front Immunol. 2015 May 15;6:231. doi: 10.3389/fimmu.2015.00231. eCollection 2015. Front Immunol. 2015. PMID: 26029216 Free PMC article. Review.
-
Cancer-Associated Fibroblast Functions as a Road-Block in Cancer Therapy.Cancers (Basel). 2021 Oct 19;13(20):5246. doi: 10.3390/cancers13205246. Cancers (Basel). 2021. PMID: 34680395 Free PMC article. Review.
Cited by
-
The Three-Dimensional In Vitro Cell Culture Models in the Study of Oral Cancer Immune Microenvironment.Cancers (Basel). 2023 Aug 25;15(17):4266. doi: 10.3390/cancers15174266. Cancers (Basel). 2023. PMID: 37686542 Free PMC article. Review.
-
Myofibroblasts: Function, Formation, and Scope of Molecular Therapies for Skin Fibrosis.Biomolecules. 2021 Jul 23;11(8):1095. doi: 10.3390/biom11081095. Biomolecules. 2021. PMID: 34439762 Free PMC article. Review.
-
YAP/TAZ Mediate TGFβ2-Induced Schlemm's Canal Cell Dysfunction.Invest Ophthalmol Vis Sci. 2022 Nov 1;63(12):15. doi: 10.1167/iovs.63.12.15. Invest Ophthalmol Vis Sci. 2022. PMID: 36350617 Free PMC article.
-
In Vitro 3D Cultures to Model the Tumor Microenvironment.Cancers (Basel). 2021 Jun 13;13(12):2970. doi: 10.3390/cancers13122970. Cancers (Basel). 2021. PMID: 34199324 Free PMC article. Review.
-
Differential Expression Profiles of Cell-to-Matrix-Related Molecules in Adrenal Cortical Tumors: Diagnostic and Prognostic Implications.J Pers Med. 2021 May 6;11(5):378. doi: 10.3390/jpm11050378. J Pers Med. 2021. PMID: 34066306 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

