An Observational Study of Acquired EGFR T790M-Dependent Resistance to EGFR-TKI Treatment in Lung Adenocarcinoma Patients in Taiwan
- PMID: 33014788
- PMCID: PMC7498675
- DOI: 10.3389/fonc.2020.01481
An Observational Study of Acquired EGFR T790M-Dependent Resistance to EGFR-TKI Treatment in Lung Adenocarcinoma Patients in Taiwan
Abstract
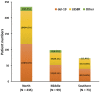
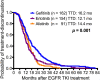
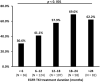
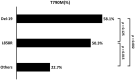
In Taiwan, epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors (EGFR-TKIs), gefitinib, erlotinib, and afatinib are served as first-line therapy for non-small lung cell cancer (NSCLC) patients with EGFR sensitizing mutations. However, the majority of patients who initially respond to EGFR-TKIs, progress through acquiring EGFR T790M mutations (T790M), which is the most common resistant mechanism. Patients with T790M gain the opportunity of subsequent treatment with third-generation EGFR-TKI, osimertinib. This study aimed to evaluate the association between prior EGFR-TKI therapy and incidence of acquired T790M resistance in lung adenocarcinoma patients who have progressed on first/second-generation EGFR-TKI therapy. This retrospective study included lung adenocarcinoma patients who had a radiographically-confirmed progressive disease under EGFR-TKI treatment and had re-biopsy samples for T790M testing from seven medical centers in Taiwan from June 2013 to December 2018. Patients harboring de novo T790M or using more than one EGFR-TKI were excluded. Of the 407 patients enrolled, the overall T790M acquisition rate was 52.8%. The patients treated with gefitinib, erlotinib or afatinib had a statistically significant difference in the T790M rates (59.9, 45.5, and 52.7%, respectively; p = 0.037) after disease progression. Patients with common baseline EGFR mutations (Del-19 and L858R) (p = 0.005) and longer treatment duration with EGFR-TKIs (p < 0.001) had higher chances of T790M acquisition. Multivariate logistic regression analysis further showed that patients with common baseline EGFR mutations, gefitinib (compared to erlotinib) administration, and longer treatment duration with EGFR-TKIs had higher T790M incidence. There was no significant difference in the incidence of acquired T790M between different re-biopsy tissue samples or complications. In conclusion, this study showed that patients who progressed from gefitinib treatment, bearing common EGFR mutations, and with longer EGFR-TKI treatment duration had increased incidence of T790M acquisition and, therefore, were suitable for subsequent osimertinib treatment.
Keywords: afatinib; epidermal growth factor receptor mutation; erlotinib; gefitinib; non-small cell lung cancer; osimertinib; tyrosine kinase inhibitor.
Copyright © 2020 Wu, Chiang, Liu, Wang, Su, Hsia, Shih and Chang.
Figures

Similar articles
-
Afatinib overcoming resistance to icotinib and osimertinib in NSCLC with leptomeningeal metastasis in patients with acquired EGFR L858R/T790M or L858R/S768I mutations: Two case reports.Heliyon. 2023 Oct 8;9(10):e20690. doi: 10.1016/j.heliyon.2023.e20690. eCollection 2023 Oct. Heliyon. 2023. PMID: 37860534 Free PMC article.
-
Clinical outcomes and secondary epidermal growth factor receptor (EGFR) T790M mutation among first-line gefitinib, erlotinib and afatinib-treated non-small cell lung cancer patients with activating EGFR mutations.Int J Cancer. 2019 Jun 1;144(11):2887-2896. doi: 10.1002/ijc.32025. Epub 2019 Jan 5. Int J Cancer. 2019. PMID: 30485437
-
Comparison of T790M Acquisition After Treatment With First- and Second-Generation Tyrosine-Kinase Inhibitors: A Systematic Review and Network Meta-Analysis.Front Oncol. 2022 Jun 28;12:869390. doi: 10.3389/fonc.2022.869390. eCollection 2022. Front Oncol. 2022. PMID: 35837103 Free PMC article.
-
Acquisition of the T790M resistance mutation during afatinib treatment in EGFR tyrosine kinase inhibitor-naïve patients with non-small cell lung cancer harboring EGFR mutations.Oncotarget. 2017 Jul 12;8(40):68123-68130. doi: 10.18632/oncotarget.19243. eCollection 2017 Sep 15. Oncotarget. 2017. PMID: 28978102 Free PMC article.
-
Osimertinib in the treatment of non-small-cell lung cancer: design, development and place in therapy.Lung Cancer (Auckl). 2017 Aug 18;8:109-125. doi: 10.2147/LCTT.S119644. eCollection 2017. Lung Cancer (Auckl). 2017. PMID: 28860885 Free PMC article. Review.
Cited by
-
Clinical utility of repeated rebiopsy for EGFR T790M mutation detection in non-small cell lung cancer.Front Oncol. 2024 Aug 26;14:1452947. doi: 10.3389/fonc.2024.1452947. eCollection 2024. Front Oncol. 2024. PMID: 39252953 Free PMC article.
-
Epidermal growth factor receptor tyrosine kinase inhibitors for de novo T790M mutation: A retrospective study of 44 patients.Thorac Cancer. 2022 Jul;13(13):1888-1897. doi: 10.1111/1759-7714.14272. Epub 2022 May 28. Thorac Cancer. 2022. PMID: 35633141 Free PMC article.
-
Sequential treatment in advanced epidermal growth factor receptor-mutated lung adenocarcinoma patients receiving first-line bevacizumab combined with 1st/2nd-generation EGFR-tyrosine kinase inhibitors.Front Oncol. 2023 Oct 3;13:1249106. doi: 10.3389/fonc.2023.1249106. eCollection 2023. Front Oncol. 2023. PMID: 37854677 Free PMC article.
-
The Impact of Acquired EGFR T790M Mutation and EGFR Circulating Cell-Free DNA on Survival in Patients with Lung Adenocarcinoma Following EGFR-TKI Therapy.Onco Targets Ther. 2021 Jan 5;13:13425-13435. doi: 10.2147/OTT.S279540. eCollection 2020. Onco Targets Ther. 2021. PMID: 33447048 Free PMC article.
-
Impact of T790M Mutation Status on Later-Line Osimertinib Treatment in Non-Small Cell Lung Cancer Patients.Cancers (Basel). 2022 Oct 18;14(20):5095. doi: 10.3390/cancers14205095. Cancers (Basel). 2022. PMID: 36291877 Free PMC article.
References
-
- Howlader N, Noone A, Krapcho M, Miller D, Brest A, Yu M, et al. SEER Cancer Statistics Review, 1975-2016, National Cancer Institute. Bethesda: National Cancer Institute; (2019).
-
- Kris M, Johnson B, Kwiatkowski D, Iafrate A, Wistuba I, Aronson S, et al. Identification of driver mutations in tumor specimens from 1,000 patients with lung adenocarcinoma: the NCI's lung cancer mutation consortium (LCMC). J Clin Oncol. (2011) 29:CRA7506 10.1371/journal.pone.0040109 - DOI
-
- Yeh P, Chen H, Andrews J, Naser R, Pao W, Horn L. DNA-mutation inventory to refine and enhance cancer treatment (DIRECT): a catalog of clinically relevant cancer mutations to enable genome-directed anticancer therapy. Clin Cancer Res. (2013) 19:1894–901. 10.1158/1078-0432.CCR-12-1894 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

