Protective Role of Tangshen Formula on the Progression of Renal Damage in db/db Mice by TRPC6/Talin1 Pathway in Podocytes
- PMID: 33015191
- PMCID: PMC7519445
- DOI: 10.1155/2020/3634974
Protective Role of Tangshen Formula on the Progression of Renal Damage in db/db Mice by TRPC6/Talin1 Pathway in Podocytes
Abstract
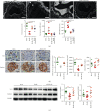
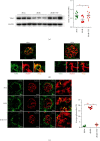
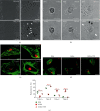
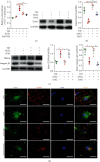
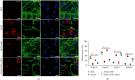
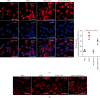
Tangshen Formula (TSF) is a Chinese Medicine formula that has been reported to alleviate proteinuria and protect renal function in humans and animals with diabetic kidney disease (DKD). However, little is known about its mechanism in improving proteinuria. The dysregulation of podocyte cell-matrix adhesion has been demonstrated to play an important role in the pathogenesis and progression of proteinuric kidney diseases including DKD. In the present study, the underlying protective mechanism of TSF on podocytes was investigated using the murine model of type 2 DKD db/db mice in vivo and advanced glycation end products (AGEs)-stimulated primary mice podocytes in vitro. Results revealed that TSF treatment could significantly mitigate reduction of podocyte numbers and foot process effacement, reduce proteinuria, and protect renal function in db/db mice. There was a significant increase in expression of transient receptor potential canonical channel 6 (TRPC6) and a decrease in expression of talin1 in podocytes of db/db mice. The results of AGEs-stimulated primary mice podocytes showed increased cell migration and actin-cytoskeleton rearrangement. Moreover, primary mice podocytes stimulated by AGEs displayed an increase in TRPC6-dependent Ca2+ influx, a loss of talin1, and translocation of nuclear factor of activated T cell (NFATC) 2. These dysregulations in mice primary podocytes stimulated by AGEs could be significantly attenuated after TSF treatment. 1-Oleoyl-2-acetyl-sn-glycerol (OAG), a TRPC6 agonist, blocked the protective role of TSF on podocyte cell-matrix adherence. In conclusion, TSF could protect podocytes from injury and reduce proteinuria in DKD, which may be mediated by the regulation of the TRPC6/Talin1 pathway in podocytes.
Copyright © 2020 Qian Wang et al.
Conflict of interest statement
The authors have no conflicts of interest to declare.
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

