Quantitative Approaches for Scoring in vivo Neuronal Aggregate and Organelle Extrusion in Large Exopher Vesicles in C. elegans
- PMID: 33016946
- PMCID: PMC7805482
- DOI: 10.3791/61368
Quantitative Approaches for Scoring in vivo Neuronal Aggregate and Organelle Extrusion in Large Exopher Vesicles in C. elegans
Abstract
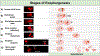
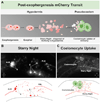
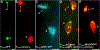
Toxicity of misfolded proteins and mitochondrial dysfunction are pivotal factors that promote age-associated functional neuronal decline and neurodegenerative disease across species. Although these neurotoxic challenges have long been considered to be cell-intrinsic, considerable evidence now supports that misfolded human disease proteins originating in one neuron can appear in neighboring cells, a phenomenon proposed to promote pathology spread in human neurodegenerative disease. C. elegans adult neurons that express aggregating proteins can extrude large (~4 µm) membrane-surrounded vesicles that can include the aggregated protein, mitochondria, and lysosomes. These large vesicles are called "exophers" and are distinct from exosomes (which are about 100x smaller and have different biogenesis). Throwing out cellular debris in exophers may occur by a conserved mechanism that constitutes a fundamental, but formerly unrecognized, branch of neuronal proteostasis and mitochondrial quality control, relevant to processes by which aggregates spread in human neurodegenerative diseases. While exophers have been mostly studied in animals that express high copy transgenic mCherry within touch neurons, these protocols are equally useful in the study of exophergenesis using fluorescently tagged organelles or other proteins of interest in various classes of neurons. Described here are the physical features of C. elegans exophers, strategies for their detection, identification criteria, optimal timing for quantitation, and animal growth protocols that control for stresses that can modulate exopher production levels. Together, details of protocols outlined here should serve to establish a standard for quantitative analysis of exophers across laboratories. This document seeks to serve as a resource in the field for laboratories seeking to elaborate molecular mechanisms by which exophers are produced and by which exophers are reacted to by neighboring and distant cells.
Figures




References
-
- Stahl PD, Raposo G Extracellular Vesicles: Exosomes and Microvesicles, Integrators of Homeostasis. Physiology (Bethesda, Md.). 34 (3), 169–177 (2019). - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
