Safety, Antitumor Activity, and Pharmacokinetics of Toripalimab, a Programmed Cell Death 1 Inhibitor, in Patients With Advanced Non-Small Cell Lung Cancer: A Phase 1 Trial
- PMID: 33017026
- PMCID: PMC7536589
- DOI: 10.1001/jamanetworkopen.2020.13770
Safety, Antitumor Activity, and Pharmacokinetics of Toripalimab, a Programmed Cell Death 1 Inhibitor, in Patients With Advanced Non-Small Cell Lung Cancer: A Phase 1 Trial
Abstract
Importance: Programmed cell death 1 (PD-1) antibodies have shown substantial survival benefit in patients with advanced non-small cell lung cancer (NSCLC). Toripalimab is a promising and practicable PD-1 antibody; however, its performance in NSCLC has not been established.
Objectives: To assess the safety, antitumor activity, and pharmacokinetics of toripalimab in patients with advanced NSCLC and to evaluate the utility of JS311, a novel PD ligand 1 (PD-L1) immunohistochemistry (IHC) assay.
Design, setting, and participants: This single-arm open-label phase 1 trial enrolled 41 patients with advanced NSCLC that had progressed after at least 3 lines of therapy between September 21, 2017, and June 5, 2018, with a median (interquartile range) follow-up of 14.9 (3.2-22.5) months and included a cohort study comparing JS311 with other PD-L1 IHC assays that included 280 NSCLC specimens collected from January 1, 2016, to May 21, 2018. Data collection was conducted from September 21, 2017, to September 27, 2019, and analysis was conducted from September 27, 2019, to December 30, 2019.
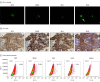
Exposure: Enrolled patients were administered a single dose of toripalimab, under 2 manufacturing processes and scales (200 L and 500 L), for safety and pharmacokinetic analysis within 28 days, followed by subsequent multidose infusions every 2 weeks. PD-L1 expression was determined by IHC with JS311, comparing its results with results from 22C3, 28-8, and SP263 simultaneously.
Main outcomes and measures: Progression-free survival (PFS) and overall survival (OS), estimated by Kaplan-Meier curves, and continuous variables compared by t test or Mann-Whitney test. Correlations between PD-L1 IHC antibodies were evaluated by Spearman correlation test.
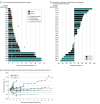
Results: A total of 41 patients (29 [70.7%] men) with a median (interquartile range) age of 59 (53 to 63) years who experienced disease progression following chemotherapy were included. The most common treatment-related adverse events were rash (6 [14.6%]), increased amylase level (5 [12.2%]), and increased aspartate aminotransferase level (5 [12.2%]). In 35 patients included in the pharmacokinetic analysis, drug exposure and area under curve after 1 dose was similar under both manufacturing processes and scales (mean [SD] for 200-L group: 12 465.28 [4128.17] hour × μg/mL; for 500-L group: 12 331.42 [2472.58] hour × μg/ml). In 28 patients included in the response and survival analysis, the median PFS and OS were 2.8 (95% CI, 2.7 to 4.6) months and 13.8 months (95% CI, 10.0 months to not reached), respectively. Stratified by PD-L1 tumor proportion score of at least 50%, 1% to 49%, and less 1%, median PFS rates were 11.2 months (95% CI, 2.3 months to not evaluable), 2.3 (95% CI, 1.7 to 2.7) months, and 2.8 (95% CI, 2.7 to 4.6) months, respectively. A total of 4 anti-PD-L1 IHC antibodies were compared during PD-L1 staining, using 280 NSCLC specimens. The consistency rates between the 4 antibodies were 80.8% to 89.5% (ρ, 0.619 to 0.790) and 93.3% to 95.5% (ρ, 0.691 to 0.773), with PD-L1 tumor proportion scores of 1% and 50% as cut points, respectively.
Conclusions and relevance: In this study, toripalimab exhibited encouraging antitumor activity and manageable safety profiles among patients with heavily treated NSCLC. The novel PD-L1 IHC antibody JS311 was highly consistent with previously verified PD-L1 IHC assays.
Conflict of interest statement
Figures


References
-
- Fehrenbacher L, Spira A, Ballinger M, et al. ; POPLAR Study Group . Atezolizumab versus docetaxel for patients with previously treated non–small cell lung cancer (POPLAR): a multicentre, open-label, phase 2 randomised controlled trial. Lancet. 2016;387(10030):1837-1846. doi: 10.1016/S0140-6736(16)00587-0 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

