c-di-AMP Accumulation Impairs Muropeptide Synthesis in Listeria monocytogenes
- PMID: 33020220
- PMCID: PMC7685554
- DOI: 10.1128/JB.00307-20
c-di-AMP Accumulation Impairs Muropeptide Synthesis in Listeria monocytogenes
Abstract
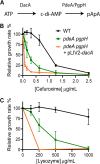
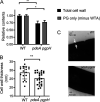
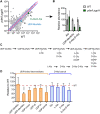
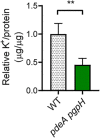
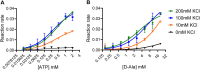
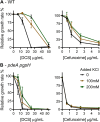
Cyclic di-AMP (c-di-AMP) is an essential and ubiquitous second messenger among bacteria. c-di-AMP regulates many cellular pathways through direct binding to several molecular targets in bacterial cells. c-di-AMP depletion is well known to destabilize the bacterial cell wall, resulting in increased bacteriolysis and enhanced susceptibility to cell wall targeting antibiotics. Using the human pathogen Listeria monocytogenes as a model, we found that c-di-AMP accumulation also impaired cell envelope integrity. An L. monocytogenes mutant deleted for c-di-AMP phosphodiesterases (pdeA pgpH mutant) exhibited a 4-fold increase in c-di-AMP levels and several cell wall defects. For instance, the pdeA pgpH mutant was defective for the synthesis of peptidoglycan muropeptides and was susceptible to cell wall-targeting antimicrobials. Among different muropeptide precursors, we found that the pdeA pgpH strain was particularly impaired in the synthesis of d-Ala-d-Ala, which is required to complete the pentapeptide stem associated with UDP-N-acetylmuramic acid (MurNAc). This was consistent with an increased sensitivity to d-cycloserine, which inhibits the d-alanine branch of peptidoglycan synthesis. Finally, upon examining d-Ala:d-Ala ligase (Ddl), which catalyzes the conversion of d-Ala to d-Ala-d-Ala, we found that its activity was activated by K+ Based on previous reports that c-di-AMP inhibits K+ uptake, we propose that c-di-AMP accumulation impairs peptidoglycan synthesis, partially through the deprivation of cytoplasmic K+ levels, which are required for cell wall-synthetic enzymes.IMPORTANCE The bacterial second messenger c-di-AMP is produced by a large number of bacteria and conditionally essential to many species. Conversely, c-di-AMP accumulation is also toxic to bacterial physiology and pathogenesis, but its mechanisms are largely undefined. We found that in Listeria monocytogenes, elevated c-di-AMP levels diminished muropeptide synthesis and increased susceptibility to cell wall-targeting antimicrobials. Cell wall defects might be an important mechanism for attenuated virulence in bacteria with high c-di-AMP levels.
Keywords: Listeria monocytogenes; c-di-AMP; peptidoglycan.
Copyright © 2020 American Society for Microbiology.
Figures

Similar articles
-
NrnA Is a Linear Dinucleotide Phosphodiesterase with Limited Function in Cyclic Dinucleotide Metabolism in Listeria monocytogenes.J Bacteriol. 2022 Jan 18;204(1):e0020621. doi: 10.1128/JB.00206-21. Epub 2021 Oct 18. J Bacteriol. 2022. PMID: 34662239 Free PMC article.
-
C-di-AMP accumulation disrupts glutathione metabolism in Listeria monocytogenes.Infect Immun. 2024 Dec 10;92(12):e0044024. doi: 10.1128/iai.00440-24. Epub 2024 Nov 19. Infect Immun. 2024. PMID: 39560402 Free PMC article.
-
Cyclic di-AMP is critical for Listeria monocytogenes growth, cell wall homeostasis, and establishment of infection.mBio. 2013 May 28;4(3):e00282-13. doi: 10.1128/mBio.00282-13. mBio. 2013. PMID: 23716572 Free PMC article.
-
A Delicate Connection: c-di-AMP Affects Cell Integrity by Controlling Osmolyte Transport.Trends Microbiol. 2018 Mar;26(3):175-185. doi: 10.1016/j.tim.2017.09.003. Epub 2017 Sep 28. Trends Microbiol. 2018. PMID: 28965724 Review.
-
Too much of a good thing: regulated depletion of c-di-AMP in the bacterial cytoplasm.Curr Opin Microbiol. 2016 Apr;30:22-29. doi: 10.1016/j.mib.2015.12.007. Epub 2016 Jan 7. Curr Opin Microbiol. 2016. PMID: 26773214 Free PMC article. Review.
Cited by
-
Coordinated regulation of osmotic imbalance by c-di-AMP shapes ß-lactam tolerance in Group B Streptococcus.Microlife. 2024 Jun 12;5:uqae014. doi: 10.1093/femsml/uqae014. eCollection 2024. Microlife. 2024. PMID: 38993744 Free PMC article.
-
Cyclic di-AMP, a multifaceted regulator of central metabolism and osmolyte homeostasis in Listeria monocytogenes.Microlife. 2023 Jan 28;4:uqad005. doi: 10.1093/femsml/uqad005. eCollection 2023. Microlife. 2023. PMID: 37223746 Free PMC article. Review.
-
NrnA Is a Linear Dinucleotide Phosphodiesterase with Limited Function in Cyclic Dinucleotide Metabolism in Listeria monocytogenes.J Bacteriol. 2022 Jan 18;204(1):e0020621. doi: 10.1128/JB.00206-21. Epub 2021 Oct 18. J Bacteriol. 2022. PMID: 34662239 Free PMC article.
-
C-di-AMP accumulation disrupts glutathione metabolism in Listeria monocytogenes.Infect Immun. 2024 Dec 10;92(12):e0044024. doi: 10.1128/iai.00440-24. Epub 2024 Nov 19. Infect Immun. 2024. PMID: 39560402 Free PMC article.
-
Structural basis of KdpD histidine kinase binding to the second messenger c-di-AMP.J Biol Chem. 2021 Jan-Jun;296:100771. doi: 10.1016/j.jbc.2021.100771. Epub 2021 May 11. J Biol Chem. 2021. PMID: 33989637 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

