Fluctuation in O-GlcNAcylation inactivates STIM1 to reduce store-operated calcium ion entry via down-regulation of Ser621 phosphorylation
- PMID: 33023909
- PMCID: PMC7863906
- DOI: 10.1074/jbc.RA120.014271
Fluctuation in O-GlcNAcylation inactivates STIM1 to reduce store-operated calcium ion entry via down-regulation of Ser621 phosphorylation
Abstract
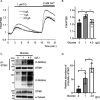
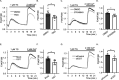
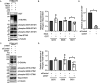
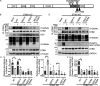
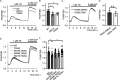
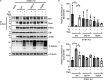
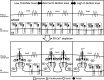
Stromal interaction molecule 1 (STIM1) plays a pivotal role in store-operated Ca2+ entry (SOCE), an essential mechanism in cellular calcium signaling and in maintaining cellular calcium balance. Because O-GlcNAcylation plays pivotal roles in various cellular function, we examined the effect of fluctuation in STIM1 O-GlcNAcylation on SOCE activity. We found that both increase and decrease in STIM1 O-GlcNAcylation impaired SOCE activity. To determine the molecular basis, we established STIM1-knockout HEK293 (STIM1-KO-HEK) cells using the CRISPR/Cas9 system and transfected STIM1 WT (STIM1-KO-WT-HEK), S621A (STIM1-KO-S621A-HEK), or T626A (STIM1-KO-T626A-HEK) cells. Using these cells, we examined the possible O-GlcNAcylation sites of STIM1 to determine whether the sites were O-GlcNAcylated. Co-immunoprecipitation analysis revealed that Ser621 and Thr626 were O-GlcNAcylated and that Thr626 was O-GlcNAcylated in the steady state but Ser621 was not. The SOCE activity in STIM1-KO-S621A-HEK and STIM1-KO-T626A-HEK cells was lower than that in STIM1-KO-WT-HEK cells because of reduced phosphorylation at Ser621 Treatment with the O-GlcNAcase inhibitor Thiamet G or O-GlcNAc transferase (OGT) transfection, which increases O-GlcNAcylation, reduced SOCE activity, whereas treatment with the OGT inhibitor ST045849 or siOGT transfection, which decreases O-GlcNAcylation, also reduced SOCE activity. Decrease in SOCE activity due to increase and decrease in O-GlcNAcylation was attributable to reduced phosphorylation at Ser621 These data suggest that both decrease in O-GlcNAcylation at Thr626 and increase in O-GlcNAcylation at Ser621 in STIM1 lead to impairment of SOCE activity through decrease in Ser621 phosphorylation. Targeting STIM1 O-GlcNAcylation could provide a promising treatment option for the related diseases, such as neurodegenerative diseases.
Keywords: O-GlcNAcylation; O-linked N-acetylglucosamine (O-GlcNAc) transferase (OGT); calcium; phosphorylation; protein phosphorylation; store-operated Ca2+ entry; store-operated calcium entry (SOCE); stromal interaction molecule 1 (STIM1).
© 2020 Nomura et al.
Conflict of interest statement
Conflict of interest—The authors declare that they have no conflicts of interest with the contents of this article.
Figures

References
-
- Holt, G. D., Haltiwanger, R. S., Torres, C. R., and Hart, G. W. (1987) Erythrocytes contain cytoplasmic glycoproteins: O-linked GlcNAc on Band 4.1. J. Biol. Chem. 262, 14847–14850 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

