Rolling circle amplification: A high fidelity and efficient alternative to plasmid preparation for the rescue of infectious clones
- PMID: 33032077
- PMCID: PMC7521378
- DOI: 10.1016/j.virol.2020.08.016
Rolling circle amplification: A high fidelity and efficient alternative to plasmid preparation for the rescue of infectious clones
Abstract
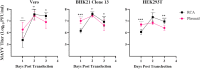
Alphaviruses (genus Alphavirus; family Togaviridae) are a medically relevant family of viruses that include chikungunya virus and Mayaro virus. Infectious cDNA clones of these viruses are necessary molecular tools to understand viral biology. Traditionally, rescuing virus from an infectious cDNA clone requires propagating plasmids in bacteria, which can result in mutations in the viral genome due to bacterial toxicity or recombination and requires specialized equipment and knowledge to propagate the bacteria. Here, we present an alternative- rolling circle amplification (RCA), an in vitro technology. We demonstrate that the viral yield of transfected RCA product is comparable to midiprepped plasmid, albeit with a slight delay in kinetics. RCA, however, is cheaper and less time-consuming. Further, sequential RCA did not introduce mutations into the viral genome, subverting the need for glycerol stocks and retransformation. These results indicate that RCA is a viable alternative to traditional plasmid-based approaches to viral rescue.
Keywords: Alphaviruses; Bacteria-free; Infectious clone; Molecular virology; Rolling circle amplification; Viral rescue.
Copyright © 2020 Elsevier Inc. All rights reserved.
Conflict of interest statement
There are no known conflicts of interest.
Figures



Similar articles
-
Direct retransformation of yeast with plasmid DNA isolated from single yeast colonies using rolling circle amplification.Biotechniques. 2003 Oct;35(4):774-6, 778-9. doi: 10.2144/03354st08. Biotechniques. 2003. PMID: 14579743
-
A sequence-independent strategy for detection and cloning of circular DNA virus genomes by using multiply primed rolling-circle amplification.J Virol. 2004 May;78(10):4993-8. doi: 10.1128/jvi.78.10.4993-4998.2004. J Virol. 2004. PMID: 15113879 Free PMC article.
-
Cloning of Complete Geminivirus Genomes by Rolling Circle Amplification (RCA).Methods Mol Biol. 2025;2912:11-18. doi: 10.1007/978-1-0716-4454-6_3. Methods Mol Biol. 2025. PMID: 40064769
-
Recent advances in rolling circle amplification-based biosensing strategies-A review.Anal Chim Acta. 2021 Mar 1;1148:238187. doi: 10.1016/j.aca.2020.12.062. Epub 2020 Dec 31. Anal Chim Acta. 2021. PMID: 33516384 Review.
-
Rolling circle amplification as isothermal gene amplification in molecular diagnostics.Biochip J. 2016;10(4):262-271. doi: 10.1007/s13206-016-0402-6. Epub 2016 Jul 29. Biochip J. 2016. PMID: 32226587 Free PMC article. Review.
Cited by
-
In vitro one-pot construction of influenza viral genomes for virus particle synthesis based on reverse genetics system.PLoS One. 2024 Nov 8;19(11):e0312776. doi: 10.1371/journal.pone.0312776. eCollection 2024. PLoS One. 2024. PMID: 39514509 Free PMC article.
-
RAP: A Novel Approach to the Rapid and Highly Sensitive Detection of Respiratory Viruses.Front Bioeng Biotechnol. 2021 Nov 5;9:766411. doi: 10.3389/fbioe.2021.766411. eCollection 2021. Front Bioeng Biotechnol. 2021. PMID: 34805120 Free PMC article. Review.
-
An in vitro workflow to create and modify infectious clones using replication cycle reaction.Virology. 2023 Aug;585:109-116. doi: 10.1016/j.virol.2023.05.013. Epub 2023 Jun 13. Virology. 2023. PMID: 37331111 Free PMC article.
-
Replication in the presence of dengue convalescent serum impacts Zika virus neutralization sensitivity and fitness.Front Cell Infect Microbiol. 2023 Mar 9;13:1130749. doi: 10.3389/fcimb.2023.1130749. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 36968111 Free PMC article.
-
The E2 glycoprotein holds key residues for Mayaro virus adaptation to the urban Aedes aegypti mosquito.PLoS Pathog. 2023 Apr 5;19(4):e1010491. doi: 10.1371/journal.ppat.1010491. eCollection 2023 Apr. PLoS Pathog. 2023. PMID: 37018377 Free PMC article.
References
-
- Bates T. Cold Spring Harbor Laboratory; 2020. Development and Characterization of Infectious Clones of Two Strains of Usutu Virus. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

