Vedolizumab treatment across antiretroviral treatment interruption in chronic HIV infection: the HAVARTI protocol for a pilot dose-ranging clinical trial to assess safety, tolerance, immunological and virological activity
- PMID: 33033101
- PMCID: PMC7545629
- DOI: 10.1136/bmjopen-2020-041359
Vedolizumab treatment across antiretroviral treatment interruption in chronic HIV infection: the HAVARTI protocol for a pilot dose-ranging clinical trial to assess safety, tolerance, immunological and virological activity
Abstract
Introduction: Continuous antiretroviral therapy (ART) suppresses HIV plasma viral load (pVL) to very low levels, which allows for some immune recovery. Discontinuation of ART leads to pVL rebound from reservoirs of persistence and latency, and progressive immunodeficiency. One promising but controversial strategy targeting CD4+ T lymphocytes with a monoclonal antibody (mAb) against α4β7 integrin has shown promise through sustained virological remission of pVL (SVR) in SIV239-infected rhesus macaques. We propose to assess the safety and tolerability of vedolizumab, a licensed humanised mAb against human α4β7 integrin, in healthy HIV-infected adults on ART. This study will also assess, by analytical treatment interruption (ATI), whether vedolizumab treatment can induce SVR beyond ART and vedolizumab treatment.
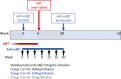
Methods and analysis: The HIV-ART-vedolizumab-ATI (HAVARTI) trial is a single-arm, dose-ranging pilot trial in healthy HIV-positive adult volunteers receiving ART. Twelve consenting persons will be enrolled in sequential groups of 4 to each serial dosing vedolizumab regimen (300 mg, 150 mg, 75 mg). The primary outcomes are: (1) to assess the safety and tolerability of seven serial infusions of vedolizumab at each of three doses; (2) to identify the immunovirological measures, including pVL and T-cell kinetics, that characterise HIV/ART cases before, during, after vedolizumab treatment and ATI; and (3) to seek SVR of pVL after ATI. Secondary outcomes will include immune reconstitution and pVL suppression as well as immune reconstitution and long-term safety following re-initiation of ART in the absence of SVR.
Ethics and dissemination: The study protocol was approved by the Ottawa Health Science Network-REB and by the Health Canada Therapeutic Products Directorate. A Data Safety Monitor will review safety information at regular intervals. The final manuscript will be submitted to an open access journal within a year of study completion.
Trial registration number: ClinicalTrials.gov NCT03147859; https://clinicaltrials.gov/ct2/show/NCT03147859.
Keywords: HIV & AIDS; clinical trials; immunology; inflammatory bowel disease.
© Author(s) (or their employer(s)) 2020. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: None declared.
Figures
References
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
