ESHRE PGT Consortium data collection XVI-XVIII: cycles from 2013 to 2015
- PMID: 33033756
- PMCID: PMC7532546
- DOI: 10.1093/hropen/hoaa043
ESHRE PGT Consortium data collection XVI-XVIII: cycles from 2013 to 2015
Abstract
Study question: What are the trends and developments in preimplantation genetic testing (PGT) in 2013-2015 as compared to previous years?
Summary answer: The main trends observed in the retrospective data collections 2013-2015, representing valuable data on PGT activity in (mainly) Europe, are the increased application of trophectoderm biopsy at the cost of cleavage stage biopsy and the continuing expansion of comprehensive testing technology in PGT for chromosomal structural rearrangements and for aneuploidies (PGT-SR and PGT-A).
What is known already: Since it was established in 1997, the ESHRE PGT Consortium has been collecting data from international PGT centres. To date, 15 data sets and an overview of the first 10 years of data collections have been published.
Study design size duration: Collection of (mainly) European data by the PGT Consortium for ESHRE. The data for PGT cycles performed between 1 January 2013 and 31 December 2015 were provided by participating centres on a voluntary basis. For the collection of cycle, pregnancy and baby data, separate, pre-designed MS Excel tables were used.
Participants/materials setting methods: Data were submitted by 59, 60 and 59 centres respectively for 2013, 2014 and 2015 (full PGT Consortium members). Records with incomplete or inconsistent data were excluded from the calculations. Corrections, calculations, figures and tables were made by expert co-authors.
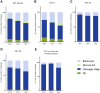
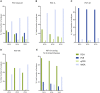
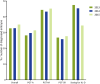
Main results and the role of chance: For data collection XVI/XVII/XVIII, 59/60/59 centres reported data on 8164/9769/11 120 cycles with oocyte retrieval: 5020/6278/7155 cycles for PGT-A, 2026/2243/2661 cycles for PGT for monogenic/single gene defects, 1039/1189/1231 cycles for PGT-SR and 79/59/73 cycles for sexing for X-linked diseases. From 2013 until 2015, the uptake of biopsy at the blastocyst stage was mainly observed in cycles for PGT-A (from 23% to 36%) and PGT-SR (from 22% to 36%), alongside the increased application of comprehensive testing technology (from 66% to 75% in PGT-A and from 36% to 58% in PGT-SR).
Limitations reasons for caution: The findings apply to the 59/60/59 participating centres and may not represent worldwide trends in PGT. Data were collected retrospectively and no details of the follow-up on PGT pregnancies and babies born were provided.
Wider implications of the findings: Being the largest data collection on PGT worldwide, detailed information about ongoing developments in the field is provided.
Study funding/competing interests: The study has no external funding and all costs are covered by ESHRE. There are no competing interests declared.
Trial registration number: N/A.
Keywords: PGD; PGT; aneuploidy; data collection; human embryo; monogenic disorders; preimplantation genetic testing; registry; structural rearrangements.
© The Author(s) 2020. Published by Oxford University Press on behalf of European Society of Human Reproduction and Embryology.
Figures



References
-
- De Rycke M, Belva F, Goossens V, Moutou C, SenGupta SB, Traeger-Synodinos J, Coonen E. ESHRE PGD Consortium data collection XIII: cycles from January to December 2010 with pregnancy follow-up to October 2011. Hum Reprod 2015;30:1763–1789. - PubMed
-
- De Rycke M, Goossens V, Kokkali G, Coonen E, Moutou C. ESHRE PGD Consortium data collection XIV-XV: cycles from January 2011 to December 2012 with pregnancy follow-up to October 2013. Hum Reprod 2017;32:1974–1994. - PubMed
-
- ESHRE PGD Consortium Steering Committee. ESHRE Preimplantation Genetic Diagnosis Consortium: data collection III (May 2001). Hum Reprod 2002;17:233–246. - PubMed
-
- ESHRE PGT Consortium and SIG-Embryology Biopsy Working Group, Kokkali G, Coticchio G, Bronet F, Celebi C, Cimadomo D, Goossens V, Liss J, Nunes S, Sfontouris I, Vermeulen Net al. ESHRE PGT Consortium and SIG Embryology Good practice recommendations for polar body and embryo biopsy for PGT. Hum Reprod Open 2020;2020:hoaa020. - PMC - PubMed
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
