Progenitor cell therapy for acquired pediatric nervous system injury: Traumatic brain injury and acquired sensorineural hearing loss
- PMID: 33034162
- PMCID: PMC7848325
- DOI: 10.1002/sctm.20-0026
Progenitor cell therapy for acquired pediatric nervous system injury: Traumatic brain injury and acquired sensorineural hearing loss
Abstract
While cell therapies hold remarkable promise for replacing injured cells and repairing damaged tissues, cell replacement is not the only means by which these therapies can achieve therapeutic effect. For example, recent publications show that treatment with varieties of adult, multipotent stem cells can improve outcomes in patients with neurological conditions such as traumatic brain injury and hearing loss without directly replacing damaged or lost cells. As the immune system plays a central role in injury response and tissue repair, we here suggest that multipotent stem cell therapies achieve therapeutic effect by altering the immune response to injury, thereby limiting damage due to inflammation and possibly promoting repair. These findings argue for a broader understanding of the mechanisms by which cell therapies can benefit patients.
Keywords: autologous stem cell transplantation; bone marrow; clinical translation; progenitor cells; umbilical cord blood.
© 2020 The Authors. STEM CELLS TRANSLATIONAL MEDICINE published by Wiley Periodicals LLC on behalf of AlphaMed Press.
Conflict of interest statement
The authors declared no potential conflicts of interest.
Figures

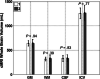

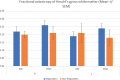
References
-
- Centers for Disease Control and Prevention . WISCARS. Leading Causes of Death Reports, National and Regional, 1999–2013; 2015. http://webappa.cdc.gov/cgi-bin/broker.exe. Accessed September 22, 2015
-
- Langlois J, Rutland‐Brown W, Thomas K. Traumatic Brain injury in the United States: Emergency Department Visits, Hospitalizations, and Deaths. Atlanta: Centers for Disease Control and Prevention, National Center for injury Prevention and Control; 2004. https://www.cdc.gov/traumaticbraininjury/tbi_ed.html. Accessed September 22, 2015.
-
- Injury Prevention and Control: Traumatic Brain Injury Centers for Disease Control and Prevention. http://www.cdc.gov/traumaticbraininjury/data/dist_death.html. Accessed September 22, 2015
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials

