Interatomic and Intermolecular Coulombic Decay
- PMID: 33035051
- PMCID: PMC7596762
- DOI: 10.1021/acs.chemrev.0c00106
Interatomic and Intermolecular Coulombic Decay
Abstract
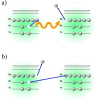
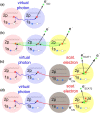
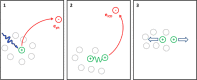
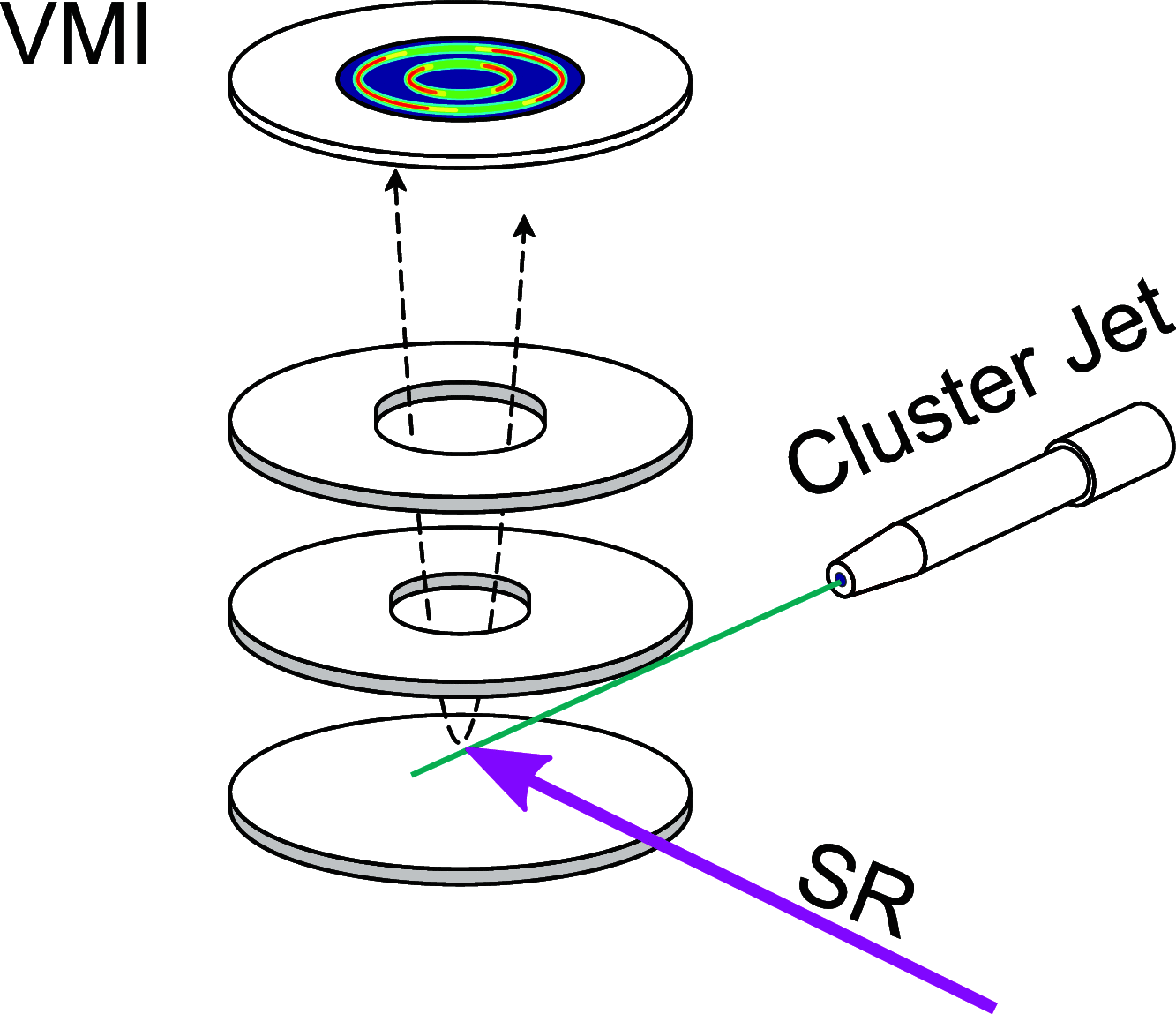
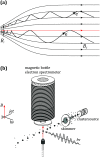
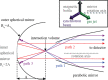
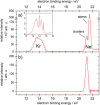
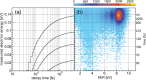
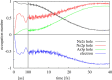
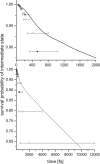
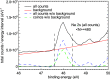
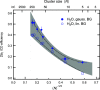
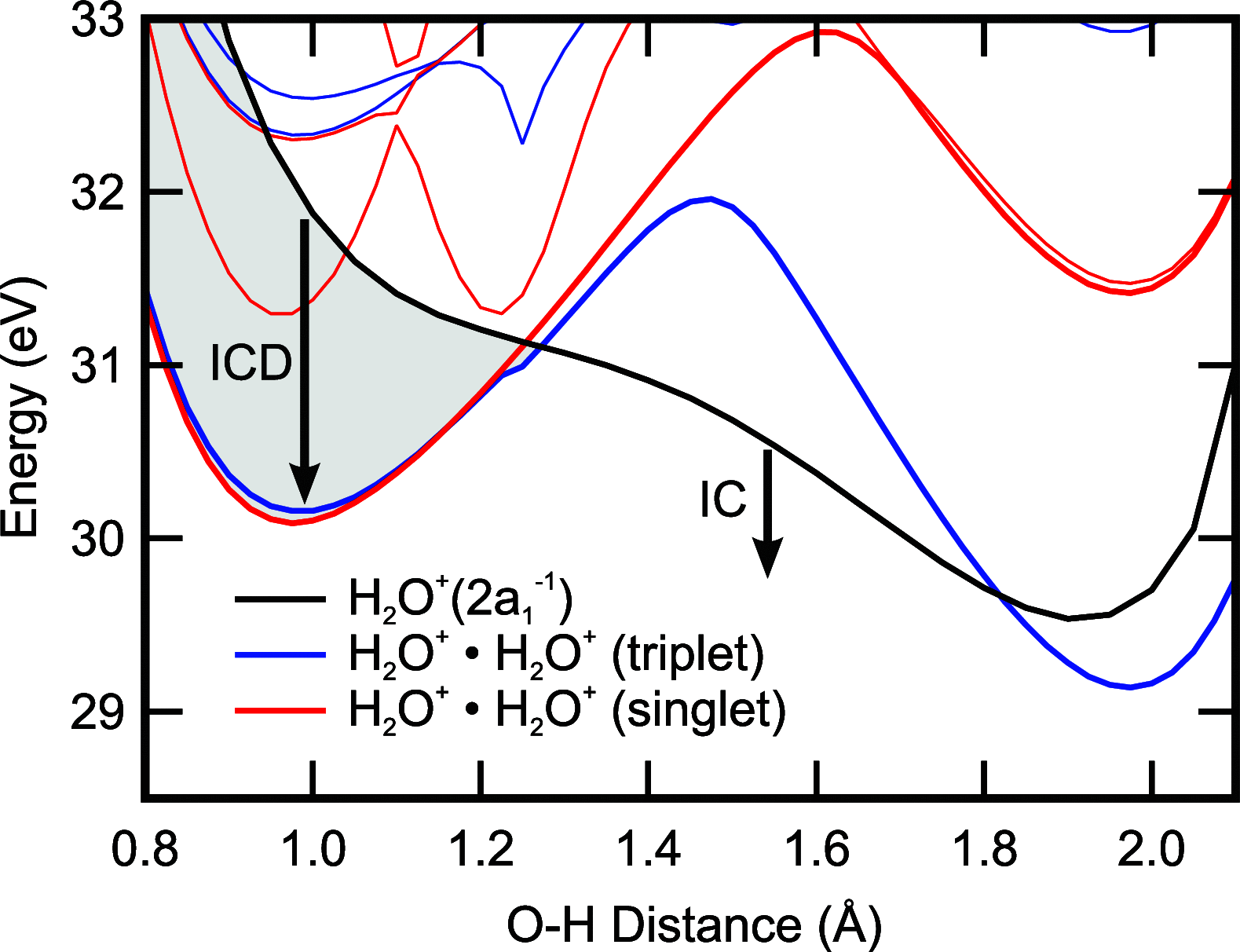
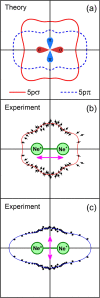
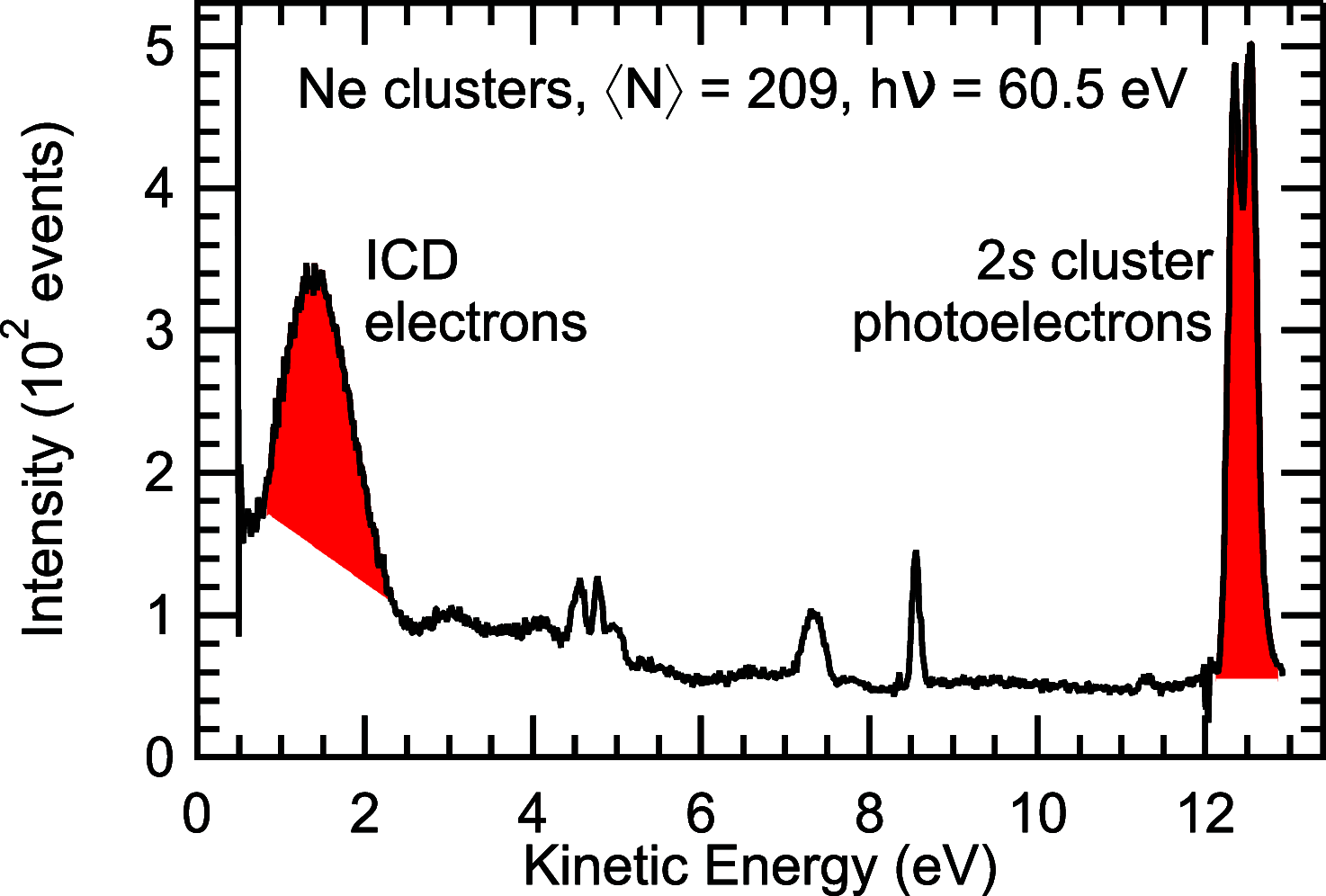
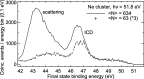
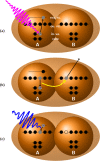
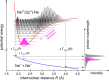
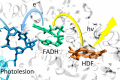
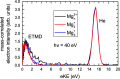
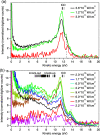
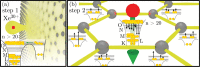
Interatomic or intermolecular Coulombic decay (ICD) is a nonlocal electronic decay mechanism occurring in weakly bound matter. In an ICD process, energy released by electronic relaxation of an excited atom or molecule leads to ionization of a neighboring one via Coulombic electron interactions. ICD has been predicted theoretically in the mid nineties of the last century, and its existence has been confirmed experimentally approximately ten years later. Since then, a number of fundamental and applied aspects have been studied in this quickly growing field of research. This review provides an introduction to ICD and draws the connection to related energy transfer and ionization processes. The theoretical approaches for the description of ICD as well as the experimental techniques developed and employed for its investigation are described. The existing body of literature on experimental and theoretical studies of ICD processes in different atomic and molecular systems is reviewed.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



















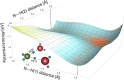

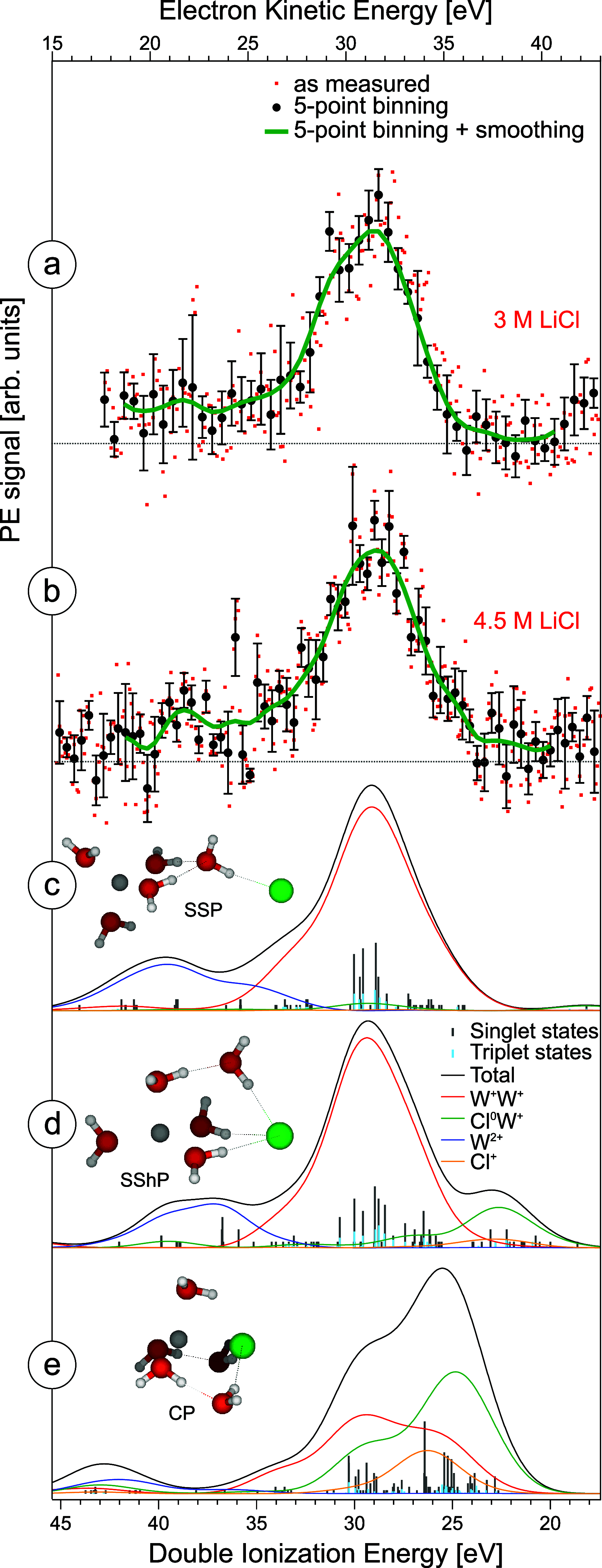
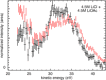



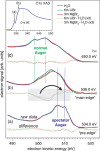



References
-
- Cederbaum L. S.; Zobeley J.; Tarantelli F. Giant InterMolecular Decay and Fragmentation of Clusters. Phys. Rev. Lett. 1997, 79, 4778–4781. 10.1103/PhysRevLett.79.4778. - DOI
-
- Öhrwall G.; Tchaplyguine M.; Lundwall M.; Feifel R.; Bergersen H.; Rander T.; Lindblad A.; Schulz J.; Peredkov S.; Barth S.; et al. Femtosecond Interatomic Coulombic Decay in Free Neon Clusters: Large Lifetime Differences between Surface and Bulk. Phys. Rev. Lett. 2004, 93, 173401.10.1103/PhysRevLett.93.173401. - DOI - PubMed
-
- Zobeley J.; Cederbaum L. S.; Tarantelli F. InterMolecular Coulombic Decay of Molecular Clusters: Identification of the Decay Mechanism Using a New Hole-Population Analysis. J. Phys. Chem. A 1999, 103, 11145–11160. 10.1021/jp992677i. - DOI
Publication types
LinkOut - more resources
Full Text Sources

