Inhibiting HMGB1-RAGE axis prevents pro-inflammatory macrophages/microglia polarization and affords neuroprotection after spinal cord injury
- PMID: 33036632
- PMCID: PMC7547440
- DOI: 10.1186/s12974-020-01973-4
Inhibiting HMGB1-RAGE axis prevents pro-inflammatory macrophages/microglia polarization and affords neuroprotection after spinal cord injury
Abstract
Background: Spinal cord injury (SCI) favors a persistent pro-inflammatory macrophages/microglia-mediated response with only a transient appearance of anti-inflammatory phenotype of immune cells. However, the mechanisms controlling this special sterile inflammation after SCI are still not fully elucidated. It is known that damage-associated molecular patterns (DAMPs) released from necrotic cells after injury can trigger severe inflammation. High mobility group box 1(HMGB1), a ubiquitously expressed DNA binding protein, is an identified DAMP, and our previous study demonstrated that reactive astrocytes could undergo necroptosis and release HMGB1 after SCI in mice. The present study aimed to explore the effects and the possible mechanism of HMGB1on macrophages/microglia polarization, as well as the neuroprotective effects by HMGB1 inhibition after SCI.
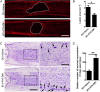
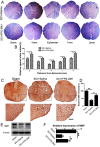
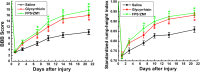
Methods: In this study, the expression and the concentration of HMGB1 was determined by qRT-PCR, ELISA, and immunohistochemistry. Glycyrrhizin was applied to inhibit HMGB1, while FPS-ZM1 to suppress receptor for advanced glycation end products (RAGE). The polarization of macrophages/microglia in vitro and in vivo was detected by qRT-PCR, immunostaining, and western blot. The lesion area was detected by GFAP staining, while neuronal survival was examined by Nissl staining. Luxol fast blue (LFB) staining, DAB staining, and western blot were adopted to evaluate the myelin loss. Basso-Beattie-Bresnahan (BBB) scoring and rump-height Index (RHI) assay was applied to evaluate locomotor functional recovery.
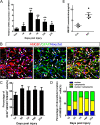
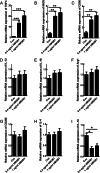
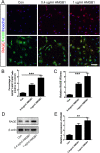
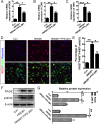
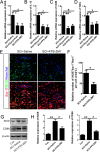
Results: Our data showed that HMGB1 can be elevated and released from necroptotic astrocytes and HMGB1 could induce pro-inflammatory microglia through the RAGE-nuclear factor-kappa B (NF-κB) pathway. We further demonstrated that inhibiting HMGB1 or RAGE effectively decreased the numbers of detrimental pro-inflammatory macrophages/microglia while increased anti-inflammatory cells after SCI. Furthermore, our data showed that inhibiting HMGB1 or RAGE significantly decreased neuronal loss and demyelination, and improved functional recovery after SCI.
Conclusions: The data implicated that HMGB1-RAGE axis contributed to the dominant pro-inflammatory macrophages/microglia-mediated pro-inflammatory response, and inhibiting this pathway afforded neuroprotection for SCI. Thus, therapies designed to modulate immune microenvironment based on this cascade might be a prospective treatment for SCI.
Keywords: HMGB1; Macrophages/microglia; Polarization; RAGE; Spinal cord injury.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Fan H, Tang HB, Shan LQ, Liu SC, Huang DG, Chen X, Chen Z, Yang M, Yin XH, Yang H, Hao DJ. Quercetin prevents necroptosis of oligodendrocytes by inhibiting macrophages/microglia polarization to M1 phenotype after spinal cord injury in rats. J Neuroinflammation. 2019;16:206. doi: 10.1186/s12974-019-1613-2. - DOI - PMC - PubMed
-
- Chen H, Ji H, Zhang M, Liu Z, Lao L, Deng C, Chen J, Zhong G. An agonist of the protective factor SIRT1 improves functional recovery and promotes neuronal survival by attenuating inflammation after spinal cord injury. J Neurosci. 2017;37:2916–2930. doi: 10.1523/JNEUROSCI.3046-16.2017. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

