Selenoprotein N-related myopathy: a retrospective natural history study to guide clinical trials
- PMID: 33037864
- PMCID: PMC7664282
- DOI: 10.1002/acn3.51218
Selenoprotein N-related myopathy: a retrospective natural history study to guide clinical trials
Abstract
Objective: To describe clinical features and disease progression of Selenoprotein N-related myopathy in a large multicenter cohort of patients.
Methods: Cross-sectional multicenter data analysis of 60 patients (53 families) with Selenoprotein N-related myopathy and single-center retrospective longitudinal analysis of 25 patients (21 families) over a median period of 5.3 years.
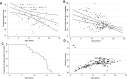
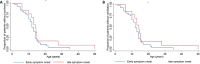
Results: The majority of patients (46/60, 77%) presented before age 2 years with hypotonia, poor head/neck control, and developmental delay. At last assessment (median age 14 years; range 2.5 to 36 years), 10/60 patients had minimal or no ambulation. Ventilatory support was initiated in 50/60 patients at a mean Forced Vital Capacity (FVC) of 38% and at a median age of 13 years. Forty-five/60 patients developed scoliosis (at median age 12.1 years) and 18 had scoliosis surgery at a median age of 13.6 years. Five children needed nasogastric feeds and/or gastrostomy. Longitudinal data analysis on 25 patients showed progressive decline of Hammersmith functional motor scores (estimated annual change -0.55 point), time to walk 10 meter, time standing from sitting, and from lying. Sixteen patients had weights < 2nd centile. The estimated change in FVC % per year was -2.04, with a 95% CI (-2.94, -1.14).
Conclusions: This comprehensive analysis of patients with Selenoprotein N-related myopathy further describes the clinical course of this rare condition. The observed functional motor and respiratory data provide evidence of the slow decline patients experience over time which is useful when considering therapeutic intervention.
© 2020 The Authors. Annals of Clinical and Translational Neurology published by Wiley Periodicals LLC on behalf of American Neurological Association.
Conflict of interest statement
Dr. Beggs serves on the scientific advisory boards of Audentes Therapeutics Inc and Dynacure Inc., has received sponsored research support from Audentes Therapeutics Inc, receives sponsored research support from Alexion Pharmaceuticals, Inc., is a paid consultant for F. Hoffmann‐La Roche, performs occasional paid consulting for Guidepoint Global Advisors and Gerson Lehrman Group, Inc., and receives research support from the US National Institutes of Health and the Muscular Dystrophy Association. There are no disclosures from other authors.
Figures



References
-
- Moghadaszadeh B, Petit N, Jaillard C, et al. Mutations in SEPN1 cause congenital muscular dystrophy with spinal rigidity and restrictive respiratory syndrome. Nat Genet 2001;29:17–18. - PubMed
-
- Ferreiro A, Ceuterick‐de Groote C, Marks JJ, et al. Desmin‐related myopathy with Mallory body‐like inclusions is caused by mutations of the selenoprotein N gene. Ann Neurol 2004;55:676–686. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
