Latent-transforming growth factor beta-binding protein-2 (LTBP-2) is required for longevity but not for development of zonular fibers
- PMID: 33039488
- PMCID: PMC8276913
- DOI: 10.1016/j.matbio.2020.10.002
Latent-transforming growth factor beta-binding protein-2 (LTBP-2) is required for longevity but not for development of zonular fibers
Abstract
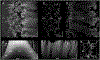
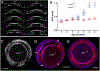
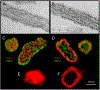
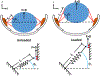
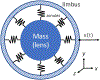
Latent-transforming growth factor beta-binding protein 2 (LTBP-2) is a major component of arterial and lung tissue and of the ciliary zonule, the system of extracellular fibers that centers and suspends the lens in the eye. LTBP-2 has been implicated previously in the development of extracellular microfibrils, although its exact role remains unclear. Here, we analyzed the three-dimensional structure of the ciliary zonule in wild type mice and used a knockout model to test the contribution of LTBP-2 to zonule structure and mechanical properties. In wild types, zonular fibers had diameters of 0.5-1.0 micrometers, with an outer layer of fibrillin-1-rich microfibrils and a core of fibrillin-2-rich microfibrils. LTBP-2 was present in both layers. The absence of LTBP-2 did not affect the number of fibers, their diameters, nor their coaxial organization. However, by two months of age, LTBP-2-depleted fibers began to rupture, and by six months, a fully penetrant ectopia lentis phenotype was present, as confirmed by in vivo imaging. To determine whether the seemingly normal fibers of young mice were compromised mechanically, we compared zonule stress/strain relationships of wild type and LTBP-2-deficient mice and developed a quasi-linear viscoelastic engineering model to analyze the resulting data. In the absence of LTBP-2, the ultimate tensile strength of the zonule was reduced by about 50%, and the viscoelastic behavior of the fibers was altered significantly. We developed a harmonic oscillator model to calculate the forces generated during saccadic eye movement. Model simulations suggested that mutant fibers are prone to failure during rapid rotation of the eyeball. Together, these data indicate that LTBP-2 is necessary for the strength and longevity of zonular fibers, but not necessarily for their formation.
Keywords: Ectopia lentis; LTBP-2; Microspherophakia; Quasi-linear viscoelastic model; Saccade; Super-resolution microscopy; Zonule.
Copyright © 2020 The Authors. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest None.
Figures

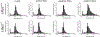
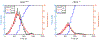
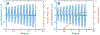



Similar articles
-
Latent TGF-β binding protein-2 is essential for the development of ciliary zonule microfibrils.Hum Mol Genet. 2014 Nov 1;23(21):5672-82. doi: 10.1093/hmg/ddu283. Epub 2014 Jun 6. Hum Mol Genet. 2014. PMID: 24908666 Free PMC article.
-
Zinn's zonule.Prog Retin Eye Res. 2021 May;82:100902. doi: 10.1016/j.preteyeres.2020.100902. Epub 2020 Sep 25. Prog Retin Eye Res. 2021. PMID: 32980533 Free PMC article. Review.
-
Latent TGF-β binding protein 2 and 4 have essential overlapping functions in microfibril development.Sci Rep. 2017 Mar 2;7:43714. doi: 10.1038/srep43714. Sci Rep. 2017. PMID: 28252045 Free PMC article.
-
Targeted deletion of fibrillin-1 in the mouse eye results in ectopia lentis and other ocular phenotypes associated with Marfan syndrome.Dis Model Mech. 2019 Jan 25;12(1):dmm037283. doi: 10.1242/dmm.037283. Dis Model Mech. 2019. PMID: 30642872 Free PMC article.
-
Zonulopathies as Genetic Disorders of the Extracellular Matrix.Genes (Basel). 2024 Dec 20;15(12):1632. doi: 10.3390/genes15121632. Genes (Basel). 2024. PMID: 39766898 Free PMC article. Review.
Cited by
-
Research progress of lens zonules.Adv Ophthalmol Pract Res. 2023 Feb 25;3(2):80-85. doi: 10.1016/j.aopr.2023.02.002. eCollection 2023 May-Jun. Adv Ophthalmol Pract Res. 2023. PMID: 37846380 Free PMC article. Review.
-
A Novel Multi-Observation System to Study the Effects of Anterior Ocular Inflammation in Zinn's Zonule Using One Specimen.Int J Mol Sci. 2023 Mar 26;24(7):6254. doi: 10.3390/ijms24076254. Int J Mol Sci. 2023. PMID: 37047225 Free PMC article.
-
Multi-organ phenotypes in mice lacking latent TGFβ binding protein 2 (LTBP2).Dev Dyn. 2024 Feb;253(2):233-254. doi: 10.1002/dvdy.651. Epub 2023 Sep 9. Dev Dyn. 2024. PMID: 37688792 Free PMC article. Review.
-
The Biomechanics of Fibrillin Microfibrils: Lessons from the Ciliary Zonule.Cells. 2024 Dec 18;13(24):2097. doi: 10.3390/cells13242097. Cells. 2024. PMID: 39768188 Free PMC article. Review.
-
Fibrillin-1-regulated miR-122 has a critical role in thoracic aortic aneurysm formation.Cell Mol Life Sci. 2022 May 23;79(6):314. doi: 10.1007/s00018-022-04337-8. Cell Mol Life Sci. 2022. PMID: 35606547 Free PMC article.
References
-
- Shipley JM, Mecham RP, Maus E, Bonadio J, Rosenbloom J, McCarthy RT, Baumann ML, Frankfater C, Segade F, Shapiro SD, Developmental expression of latent transforming growth factor beta binding protein 2 and its requirement early in mouse development, Mol Cell Biol 20(13) (2000) 4879–87. - PMC - PubMed
-
- Gibson MA, Hatzinikolas G, Davis EC, Baker E, Sutherland GR, Mecham RP, Bovine latent transforming growth factor beta 1-binding protein 2: molecular cloning, identification of tissue isoforms, and immunolocalization to elastin-associated microfibrils, Mol Cell Biol 15(12) (1995) 6932–42. - PMC - PubMed
-
- Ali M, McKibbin M, Booth A, Parry DA, Jain P, Riazuddin SA, Hejtmancik JF, Khan SN, Firasat S, Shires M, Gilmour DF, Towns K, Murphy AL, Azmanov D, Tournev I, Cherninkova S, Jafri H, Raashid Y, Toomes C, Craig J, Mackey DA, Kalaydjieva L, Riazuddin S, Inglehearn CF, Null mutations in LTBP2 cause primary congenital glaucoma, Am J Hum Genet 84(5) (2009) 664–71. - PMC - PubMed
-
- Narooie-Nejad M, Paylakhi SH, Shojaee S, Fazlali Z, Rezaei Kanavi M, Nilforushan N, Yazdani S, Babrzadeh F, Suri F, Ronaghi M, Elahi E, Paisan-Ruiz C, Loss of function mutations in the gene encoding latent transforming growth factor beta binding protein 2, LTBP2, cause primary congenital glaucoma, Hum Mol Genet 18(20) (2009) 3969–77. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

