Leveraging implementation science to improve implementation outcomes in precision medicine
- PMID: 33042394
- PMCID: PMC7540127
Leveraging implementation science to improve implementation outcomes in precision medicine
Abstract
Background and purpose: Introduction of omics technologies in clinical practice means increased use of validated biomarkers, through precision medicine (PM). Although implementation science (IS) affords an array of theoretical approaches that can potentially explain PM intervention uptake, their relevance and applicability in PM implementation has not been empirically tested. This article identifies and examines existing implementation frameworks for their applicability in PM, demonstrating how different IS theories can be used to generate testable implementation hypotheses in PM.
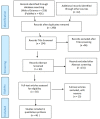
Methods: A three-step methodology was employed to search and select implementation models: a scoping search in Google Scholar produced 15 commonly used models in healthcare; a systematic search in PUBMED and Web of Science using the names of each model as keywords in search strings produced 290 publications for screening and abstraction; finally, a citation frequency search in the 3 databases produced most cited models that were included in the narrative synthesis.
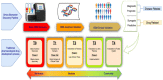
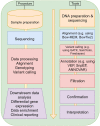
Results: Main concepts and constructs associated with each of the 15 models were identified. Four most cited frameworks in healthcare were: REAIM, CFIR, PRISM and PARiHS. Corresponding constructs were mapped and examined for potential congruence to PM. A generalized PM implementation conceptual framework was developed showing how omics biomarker uptake relates to their evidence base, patient and provider engagement and Big data capabilities of involved organizations.
Conclusion: We demonstrated how implementation complexities in PM can be addressed by explicit use of implementation theories. The work here may provide a reference for further research of empirically testing and refining the identified implementation constructs.
Keywords: Implementation science; biomarkers; genomic medicine; omics technologies; precision medicine.
AJTR Copyright © 2020.
Conflict of interest statement
None.
Figures

References
-
- Anderson C. The end of theory: the data deluge makes the scientific method obsolete. Wired Magazine. 2008;16:16–07.
-
- Zhang X. Precision medicine, personalized medicine, omics and big data: concepts and relationships. J Pharmacogenomics Pharmacoproteomics. 2015;6:e144.
-
- Eccles MP, Johnston M, Hrisos S, Francis J, Grimshaw J, Steen N, Kaner EF. Translating clinicians’ beliefs into implementation interventions (TRACII): a protocol for an intervention modeling experiment to change clinicians’ intentions to implement evidence-based practice. Implement Sci. 2007;2:27. - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
