Health care utilization and cost associated with switching biologics within the first year of biologic treatment initiation among patients with ankylosing spondylitis
- PMID: 33043820
- PMCID: PMC10394190
- DOI: 10.18553/jmcp.2020.19433
Health care utilization and cost associated with switching biologics within the first year of biologic treatment initiation among patients with ankylosing spondylitis
Abstract
Background: Costs associated with biologic switching and discontinuation can be high in chronic inflammatory diseases. Inappropriate use of medications may have cost implications for both payers and patients. Understanding of biologic utilization and switching rates is lacking among patients with ankylosing spondylitis (AS).
Objective: To compare direct costs of treatment switchers, nonswitchers, and discontinuers among patients with AS who newly initiated a biologic.
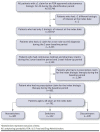
Methods: Patients aged ≥ 18 years with an AS diagnosis and ≥ 1 pharmacy claim for an FDA-approved subcutaneous biologic for AS between January 1, 2016, and December 31, 2016 (identification period) were identified from the Truven Health MarketScan Databases. At the time of biologic initiation (index date), eligible patients were continuously enrolled with medical and pharmacy claims for ≥ 1 year before (baseline period) and ≥ 1 year after the index date (follow-up period). Patients with AS were categorized into 3 mutually exclusive groups of nonswitchers (patients who remained on their index biologic), switchers (patients who had a prescription for a biologic therapy other than their index biologic), and discontinuers (patients who had gaps in prescription claims [> 120 days]) based on their biologic utilization pattern during the 1-year follow-up period. Health care costs (inflated to year 2017 costs) during follow-up were described across the 3 groups separately and by disease. Adjusted health care resource utilization and costs during 1-year follow-up were estimated, controlling for age, sex, fulltime work status, and Charlson Comorbidity Index (Deyo's modification) during the baseline period.
Results: A total of 791 patients with AS who were newly initiating a biologic were categorized as switchers (15.4%), nonswitchers (53.1%), and discontinuers (31.5%). Switchers and discontinuers had higher all-cause health care utilization than nonswitchers during the 1-year follow-up period. Switchers had higher mean total health care costs than nonswitchers ($71,280 vs. $66,573) due to increased medical ($13,897 vs. $12,043) and pharmacy costs ($57,384 vs. $54,530). Discontinuers had the lowest total health care costs ($41,179) but had the highest medical costs ($19,079). Adjusted analyses for covariates confirmed similar trends as the unadjusted analysis, where discontinuers had significantly lower total health care and pharmacy costs but had higher medical costs (with increased emergency department and outpatient visits) than nonswitchers (all P < 0.001). Switchers had increased outpatient visits and costs versus nonswitchers in the adjusted analysis (P < 0.001); small sample sizes in the switcher group may have limited the ability to detect significant trends in the adjusted analysis.
Conclusions: Biologic switching resulted in higher total health care costs than remaining on the same biologic in patients with AS. These findings highlight the clinical and economic impact of discontinuing or switching biologic therapies in patients with AS, which may inform treatment and/or formulary decision making.
Disclosures: This study was sponsored by Novartis Pharmaceuticals (East Hanover, NJ). Yi, Dai, and Park are employed by Novartis. Piao was an employee of KMK Consulting, supporting Novartis at the time of this study; Zheng is an employee of KMK Consulting and works as a consultant for Novartis. Portions of this work were presented at the 2019 AMCP Managed Care and Specialty Pharmacy Annual Meeting; March 25-28, 2019; San Diego, CA; and AMCP Nexus 2019; October 29-November 1, 2019; National Harbor, MD.
Conflict of interest statement
This study was sponsored by Novartis Pharmaceuticals (East Hanover, NJ). Yi, Dai, and Park are employed by Novartis. Piao was an employee of KMK Consulting, supporting Novartis at the time of this study; Zheng is an employee of KMK Consulting and works as a consultant for Novartis.
Portions of this work were presented at the 2019 AMCP Managed Care and Specialty Pharmacy Annual Meeting; March 25-28, 2019; San Diego, CA; and AMCP Nexus 2019; October 29-November 1, 2019; National Harbor, MD.
References
-
- Braun J, Sieper J. Ankylosing spondylitis. Lancet. 2007;369(9570):1379-90. - PubMed
-
- Sieper J, van der Heijde D, Landewe R, et al. . New criteria for inflammatory back pain in patients with chronic back pain: a real patient exercise by experts from the Assessment of SpondyloArthritis International Society (ASAS). Ann Rheum Dis. 2009;68(6):784-88. - PubMed
-
- Rudwaleit M, Jurik AG, Hermann KG, et al. . Defining active sacroiliitis on magnetic resonance imaging (MRI) for classification of axial spondyloarthritis: a consensual approach by the ASAS/OMERACT MRI group. Ann Rheum Dis. 2009;68(10):1520-7. - PubMed
-
- van der Linden S, Valkenburg HA, Cats A. Evaluation of diagnostic criteria for ankylosing spondylitis. A proposal for modification of the New York criteria. Arthritis Rheum. 1984;27(4):361-68. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials