Escherichia coli K12 Upregulates Programmed Cell Death Ligand 1 (PD-L1) Expression in Gamma Interferon-Sensitized Intestinal Epithelial Cells via the NF-κB Pathway
- PMID: 33046511
- PMCID: PMC7927934
- DOI: 10.1128/IAI.00618-20
Escherichia coli K12 Upregulates Programmed Cell Death Ligand 1 (PD-L1) Expression in Gamma Interferon-Sensitized Intestinal Epithelial Cells via the NF-κB Pathway
Abstract
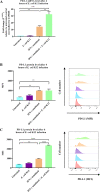
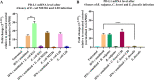
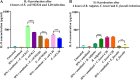
Programmed cell death ligand-1 (PD-L1) is an immune checkpoint protein which is used by tumor cells for immune evasion. PD-L1 is upregulated in inflamed intestinal tissues. The intestinal tract is colonized by millions of bacteria, most of which are commensal bacterial species. We hypothesized that under inflammatory conditions, some commensal bacterial species contribute to increased PD-L1 expression in intestinal epithelium and examined this hypothesis. Human intestinal epithelial HT-29 cells with and without interferon (IFN)-γ sensitization were incubated with six strains of four enteric bacterial species. The mRNA and protein levels of PD-L1 in HT-29 cells were examined using quantitative real-time PCR and flow cytometry, respectively. The levels of interleukin (IL)-1β, IL-18, IL-6, IL-8, and tumor necrosis factor (TNF)-α secreted by HT-29 cells were measured using enzyme-linked immunosorbent assay. Apoptosis of HT-29 cells was measured using a caspase 3/7 assay. We found that Escherichia coli K12 significantly upregulated both PD-L1 mRNA and protein in IFN-γ-sensitized HT-29 cells. E. coli K12 induced the production of IL-8 in HT-29 cells, however, IL-8 did not affect HT-29 PD-L1 expression. Inhibition of the nuclear factor-kappa B pathway significantly reduced E. coli K12-induced PD-L1 expression in HT-29 cells. The other two E. coli strains and two enteric bacterial species did not significantly affect PD-L1 expression in HT-29 cells. Enterococcus faecalis significantly inhibited PD-L1 expression due to induction of cell death. Data from this study suggest that some gut bacterial species have the potential to affect immune function under inflammatory conditions via upregulating epithelial PD-L1 expression.
Keywords: E. coli K12; IFN-γ; PD-L1; inflammation; intestinal epithelial cells.
Copyright © 2020 American Society for Microbiology.
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

