Role of opioid signaling in kidney damage during the development of salt-induced hypertension
- PMID: 33046522
- PMCID: PMC7556751
- DOI: 10.26508/lsa.202000853
Role of opioid signaling in kidney damage during the development of salt-induced hypertension
Abstract
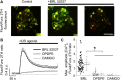
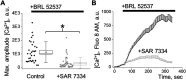
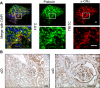
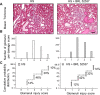
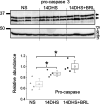
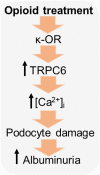
Opioid use is associated with predictors of poor cardiorenal outcomes. However, little is known about the direct impact of opioids on podocytes and renal function, especially in the context of hypertension and CKD. We hypothesize that stimulation of opioid receptors (ORs) contributes to dysregulation of intracellular calcium ([Ca2+]i) homeostasis in podocytes, thus aggravating the development of renal damage in hypertensive conditions. Herein, freshly isolated glomeruli from Dahl salt-sensitive (SS) rats and human kidneys, as well as immortalized human podocytes, were used to elucidate the contribution of specific ORs to calcium influx. Stimulation of κ-ORs, but not μ-ORs or δ-ORs, evoked a [Ca2+]i transient in podocytes, potentially through the activation of TRPC6 channels. κ-OR agonist BRL52537 was used to assess the long-term effect in SS rats fed a high-salt diet. Hypertensive rats chronically treated with BRL52537 exhibited [Ca2+]i overload in podocytes, nephrinuria, albuminuria, changes in electrolyte balance, and augmented blood pressure. These data demonstrate that the κ-OR/TRPC6 signaling directly influences podocyte calcium handling, provoking the development of kidney injury in the opioid-treated hypertensive cohort.
© 2020 Golosova et al.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures





Similar articles
-
Renal Implications of Kappa Opioid Receptor Signaling in Sprague-Dawley Rats.Function (Oxf). 2025 Aug 1;6(4):zqaf028. doi: 10.1093/function/zqaf028. Function (Oxf). 2025. PMID: 40586679 Free PMC article.
-
A NOX4/TRPC6 Pathway in Podocyte Calcium Regulation and Renal Damage in Diabetic Kidney Disease.J Am Soc Nephrol. 2018 Jul;29(7):1917-1927. doi: 10.1681/ASN.2018030280. Epub 2018 May 23. J Am Soc Nephrol. 2018. PMID: 29793963 Free PMC article.
-
Angiotensin II has acute effects on TRPC6 channels in podocytes of freshly isolated glomeruli.Kidney Int. 2014 Sep;86(3):506-14. doi: 10.1038/ki.2014.71. Epub 2014 Mar 19. Kidney Int. 2014. PMID: 24646854 Free PMC article.
-
TRPC6 channel as an emerging determinant of the podocyte injury susceptibility in kidney diseases.Am J Physiol Renal Physiol. 2015 Sep 1;309(5):F393-7. doi: 10.1152/ajprenal.00186.2015. Epub 2015 Jun 17. Am J Physiol Renal Physiol. 2015. PMID: 26084930 Free PMC article. Review.
-
Balancing calcium signals through TRPC5 and TRPC6 in podocytes.J Am Soc Nephrol. 2011 Nov;22(11):1969-80. doi: 10.1681/ASN.2011040370. Epub 2011 Oct 6. J Am Soc Nephrol. 2011. PMID: 21980113 Free PMC article. Review.
Cited by
-
Transcriptomic changes in glomeruli in response to a high salt challenge in the Dahl SS rat.Physiol Genomics. 2024 Jan 1;56(1):98-111. doi: 10.1152/physiolgenomics.00075.2023. Epub 2023 Nov 13. Physiol Genomics. 2024. PMID: 37955135 Free PMC article.
-
β-Arrestin pathway activation by selective ATR1 agonism promotes calcium influx in podocytes, leading to glomerular damage.Clin Sci (Lond). 2023 Dec 22;137(24):1789-1804. doi: 10.1042/CS20230313. Clin Sci (Lond). 2023. PMID: 38051199 Free PMC article.
-
Ion channels and channelopathies in glomeruli.Physiol Rev. 2023 Jan 1;103(1):787-854. doi: 10.1152/physrev.00013.2022. Epub 2022 Aug 25. Physiol Rev. 2023. PMID: 36007181 Free PMC article. Review.
-
The role of TRPC6 channel in chronic kidney disease.Am J Physiol Renal Physiol. 2022 Feb 1;322(2):F195-F196. doi: 10.1152/ajprenal.00455.2021. Epub 2022 Jan 17. Am J Physiol Renal Physiol. 2022. PMID: 35037467 Free PMC article. No abstract available.
-
Effect of SGLT2 inhibition on salt-induced hypertension in female Dahl SS rats.Sci Rep. 2023 Nov 6;13(1):19231. doi: 10.1038/s41598-023-46016-z. Sci Rep. 2023. PMID: 37932290 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
