Degradation and detoxification of azo dyes with recombinant ligninolytic enzymes from Aspergillus sp. with secretory overexpression in Pichia pastoris
- PMID: 33047030
- PMCID: PMC7540776
- DOI: 10.1098/rsos.200688
Degradation and detoxification of azo dyes with recombinant ligninolytic enzymes from Aspergillus sp. with secretory overexpression in Pichia pastoris
Abstract
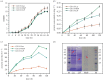
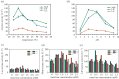
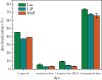
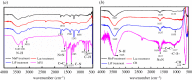
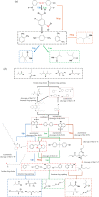
Ligninolytic enzymes, including laccase (Lac), manganese peroxidase (MnP) and lignin peroxidase (LiP), have attracted much attention in the degradation of contaminants. Genes of Lac (1827 bp), MnP (1134 bp) and LiP (1119 bp) were cloned from Aspergillus sp. TS-A, and the recombinant Lac (69 kDa), MnP (45 kDa) and LiP (35 kDa) were secretory expressed in Pichia pastoris GS115, with enzyme activities of 34, 135.12 and 103.13 U l-1, respectively. Dyes of different structures were treated via the recombinant ligninolytic enzymes under the optimal degradation conditions, and the result showed that the decolourization rate of Lac on Congo red (CR) in 5 s was 45.5%. Fourier-transform infrared spectroscopy, gas chromatography-mass spectrometry analysis and toxicity tests further proved that the ligninolytic enzymes could destroy the dyes, both those with one or more azo bonds, and the degradation products were non-toxic. Moreover, the combined ligninolytic enzymes could degrade CR more completely compared with the individual enzyme. Remarkably, besides azo dyes, ligninolytic enzymes could also degrade triphenylmethane and anthracene dyes. This suggests that ligninolytic enzymes from Aspergillus sp. TS-A have the potential for application in the treatment of contaminants.
Keywords: Aspergillus; azo dyes; degradation; detoxification; ligninolytic enzymes.
© 2020 The Authors.
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Harnessing the potential of white rot fungi and ligninolytic enzymes for efficient textile dye degradation: A comprehensive review.Water Environ Res. 2024 Jan;96(1):e10959. doi: 10.1002/wer.10959. Water Environ Res. 2024. PMID: 38204323 Review.
-
Decolourization and biodegradation of methylene blue dye by a ligninolytic enzyme-producing Bacillus thuringiensis: Degradation products and pathway.Enzyme Microb Technol. 2022 May;156:109999. doi: 10.1016/j.enzmictec.2022.109999. Epub 2022 Jan 31. Enzyme Microb Technol. 2022. PMID: 35151126
-
Lignin bioconversion based on genome mining for ligninolytic genes in Erwinia billingiae QL-Z3.Biotechnol Biofuels Bioprod. 2024 Feb 15;17(1):25. doi: 10.1186/s13068-024-02470-z. Biotechnol Biofuels Bioprod. 2024. PMID: 38360683 Free PMC article.
-
Elucidation of fungal dye-decolourizing peroxidase (DyP) and ligninolytic enzyme activities in decolourization and mineralization of azo dyes.J Appl Microbiol. 2020 Dec;129(6):1633-1643. doi: 10.1111/jam.14731. Epub 2020 Jul 9. J Appl Microbiol. 2020. PMID: 32491245
-
Ligninolytic enzymes and its mechanisms for degradation of lignocellulosic waste in environment.Heliyon. 2020 Feb 19;6(2):e03170. doi: 10.1016/j.heliyon.2020.e03170. eCollection 2020 Feb. Heliyon. 2020. PMID: 32095645 Free PMC article. Review.
Cited by
-
Improving the Properties of Laccase Through Heterologous Expression and Protein Engineering.Microorganisms. 2025 Jun 18;13(6):1422. doi: 10.3390/microorganisms13061422. Microorganisms. 2025. PMID: 40572310 Free PMC article. Review.
-
COVID-19 pandemic: A multidisciplinary perspective on the pathogenesis of a novel coronavirus from infection, immunity and pathological responses.Front Immunol. 2022 Aug 26;13:978619. doi: 10.3389/fimmu.2022.978619. eCollection 2022. Front Immunol. 2022. PMID: 36091053 Free PMC article. Review.
-
Fluorescent carbon nanodot-based rapid cell nucleus staining and application for diagnosis.J Food Drug Anal. 2024 Jun 15;32(2):239-249. doi: 10.38212/2224-6614.3503. J Food Drug Anal. 2024. PMID: 38934693 Free PMC article.
-
Advancing bioremediation: biosurfactants as catalysts for sustainable remediation.Biodegradation. 2025 Apr 16;36(3):33. doi: 10.1007/s10532-025-10128-2. Biodegradation. 2025. PMID: 40237836 Review.
-
COVID-19 and thrombosis: From bench to bedside.Trends Cardiovasc Med. 2021 Apr;31(3):143-160. doi: 10.1016/j.tcm.2020.12.004. Epub 2020 Dec 16. Trends Cardiovasc Med. 2021. PMID: 33338635 Free PMC article. Review.
References
-
- Sen SK, Raut S, Bandyopadhyay P, Raut S. 2016. Fungal decolouration and degradation of azo dyes: a review. Fungal Biol. Rev. 30, 112–133. (10.1016/j.fbr.2016.06.003) - DOI
-
- Popli S, Patel UD. 2015. Destruction of azo dyes by anaerobic–aerobic sequential biological treatment: a review. Int. J. Environ. Sci. Technol. 12, 405–420. (10.1007/s13762-014-0499-x) - DOI
-
- Kuppusamy S, Venkateswarlu K, Thavama P, Naidu R, Lee YB, Megharaj M. 2017. Quercus robur acorn peel as a novel coagulating adsorbent for cationic dye removal from aquatic ecosystems. Ecol. Eng. 101, 3–8. (10.1016/j.ecoleng.2017.01.14) - DOI
Associated data
LinkOut - more resources
Full Text Sources
Research Materials

