Exploring the Potential of Therapeutic Agents Targeted towards Mitigating the Events Associated with Amyloid-β Cascade in Alzheimer's Disease
- PMID: 33050199
- PMCID: PMC7589257
- DOI: 10.3390/ijms21207443
Exploring the Potential of Therapeutic Agents Targeted towards Mitigating the Events Associated with Amyloid-β Cascade in Alzheimer's Disease
Abstract
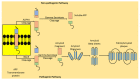
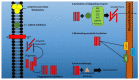
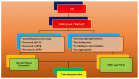
One of the most commonly occurring neurodegenerative disorders, Alzheimer's disease (AD), encompasses the loss of cognitive and memory potential, impaired learning, dementia and behavioral defects, and has been prevalent since the 1900s. The accelerating occurrence of AD is expected to reach 65.7 million by 2030. The disease results in neural atrophy and disrupted inter-neuronal connections. Amongst multiple AD pathogenesis hypotheses, the amyloid beta (Aβ) cascade is the most relevant and accepted form of the hypothesis, which suggests that Aβ monomers are formed as a result of the cleavage of amyloid precursor protein (APP), followed by the conversion of these monomers to toxic oligomers, which in turn develop β-sheets, fibrils and plaques. The review targets the events in the amyloid hypothesis and elaborates suitable therapeutic agents that function by hindering the steps of plaque formation and lowering Aβ levels in the brain. The authors discuss treatment possibilities, including the inhibition of β- and γ-secretase-mediated enzymatic cleavage of APP, the immune response generating active immunotherapy and passive immunotherapeutic approaches targeting monoclonal antibodies towards Aβ aggregates, the removal of amyloid aggregates by the activation of enzymatic pathways or the regulation of Aβ circulation, glucagon-like peptide-1 (GLP-1)-mediated curbed accumulation and the neurotoxic potential of Aβ aggregates, bapineuzumab-mediated vascular permeability alterations, statin-mediated Aβ peptide degradation, the potential role of ibuprofen and the significance of natural drugs and dyes in hindering the amyloid cascade events. Thus, the authors aim to highlight the treatment perspective, targeting the amyloid hypothesis, while simultaneously emphasizing the need to conduct further investigations, in order to provide an opportunity to neurologists to develop novel and reliable treatment therapies for the retardation of AD progression.
Keywords: Alzheimer’s disease (AD); amyloid beta (Aβ) cascade; amyloid precursor protein; immune response; toxic oligomers.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Barker W.W., Luis C.A., Kashuba A., Luis M., Harwood D.G., Loewenstein D., Waters C., Jimison P., Shepherd E., Sevush S. Relative frequencies of Alzheimer disease, Lewy body, vascular and frontotemporal dementia, and hippocampal sclerosis in the State of Florida Brain Bank. Alzheimer Dis. Assoc. Disord. 2002;16:203–212. doi: 10.1097/00002093-200210000-00001. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

